
Newsletter Subscribe
Enter your email address below and subscribe to our newsletter


Enter your email address below and subscribe to our newsletter

Lupin Limited announced the launch of Bromfenac Ophthalmic Solution, 0.075%. The product has received the green light from the United States Food and Drug Administration (U.S. FDA), marking a crucial milestone for the company’s expansion in the American pharmaceutical market.…

The European Society of Cataract and Refractive Surgeons (ESCRS) has commenced its highly anticipated Winter Meeting in Frankfurt, Germany. The event, which runs from February 15th to 18th, 2024, promises to be a hub of innovation and knowledge exchange in…

Opthea has reached a significant milestone in its quest to combat wet age-related macular degeneration (AMD) with the completion of patient enrollment in the COAST phase 3 pivotal clinical trial. The trial investigates sozinibercept (OPT-302), a promising vascular endothelial growth…
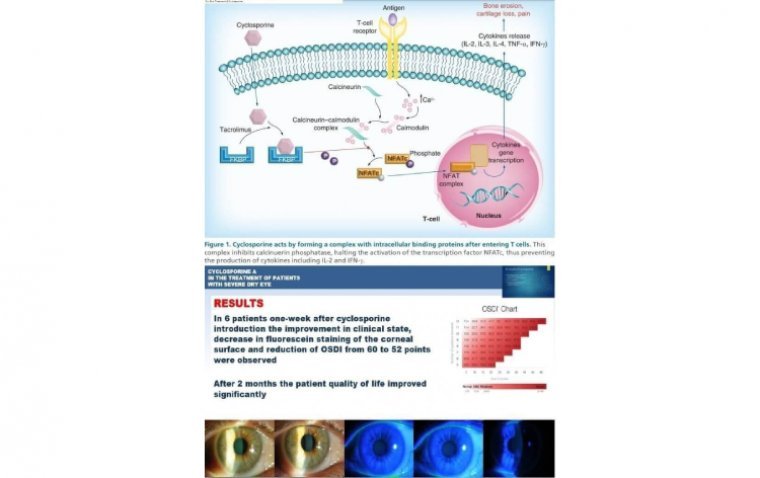
Dry eye disease (DED), also known as keratoconjunctivitis sicca (KCS), is a common disorder of the eye affecting millions of people around the world. The prevalence of DED ranges from 5% to 33% worldwide with increased prevalence among adult women…

As OBN, we had the privilege of participating in the ESCRS Congress 2023 held in Vienna, where we witnessed the remarkable efforts of various companies dedicated to enhancing patient outcomes. In this exclusive article, we present a compilation of ten…
The world of ophthalmology is constantly evolving, with innovative treatments and procedures emerging to address a wide range of eye conditions. One such cutting-edge treatment, corneal collagen cross-linking (CXL), has gained significant attention for its role in managing keratoconus, a…

The U.S. Food and Drug Administration (FDA) is warning consumers against the purchase and use of South Moon, Rebright, or FivFivGo eye drops due to the potential risk of eye infection. These products, identified as copycats, pose a danger as…
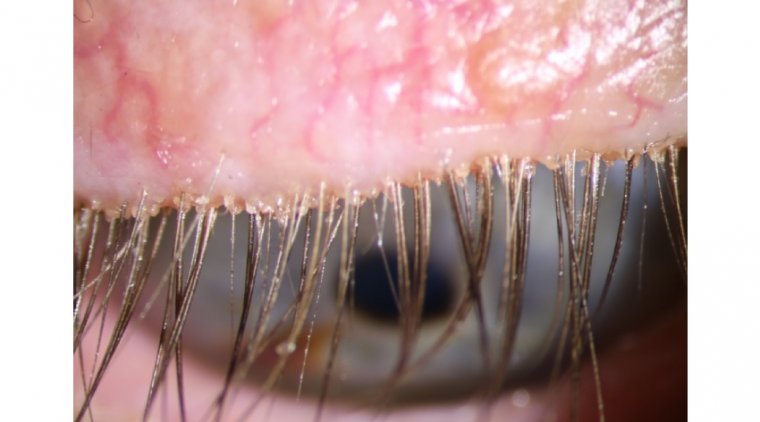
A team of investigators, led by Elizabeth Yeu, MD, from Virginia Eye Consultants in Norfolk, Virginia, reported the safety and efficacy of lotilaner ophthalmic solution, 0.25% (TP-03, Tarsus Pharmaceuticals Inc.), in treating Demodex blepharitis. Demodex mites are the most common…

Swiss-based pharmaceutical company Roche has announced the discontinuation of its oral diabetic retinopathy treatment, vicasinabin, after successfully completing Phase II clinical trials. In a recent presentation to investors, Roche confirmed its decision to halt further investment in vicasinabin, a promising…
After 30 years of dedicated work, two researchers have achieved a remarkable breakthrough by developing eye drops that can effectively reach the back of the eye. This groundbreaking development paves the way for needle-free treatments of blinding retinal diseases, such…