
Newsletter Subscribe
Enter your email address below and subscribe to our newsletter


Enter your email address below and subscribe to our newsletter
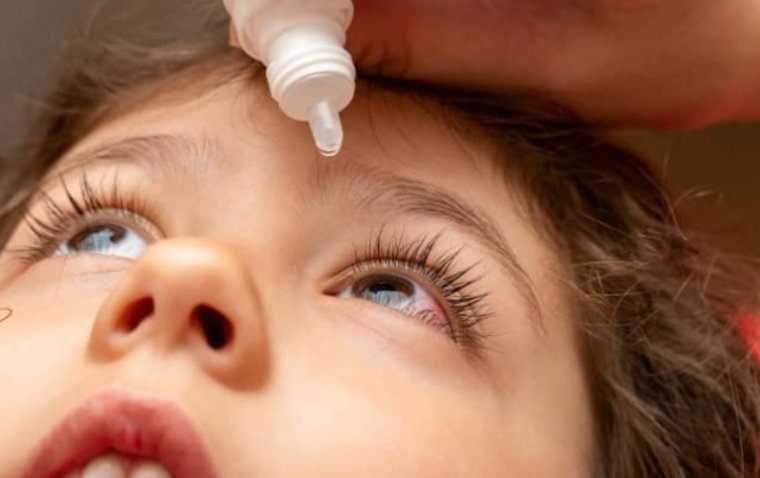
Krystal Biotech’s topical gene therapy Vyjuvek (beremagene-geperpavec) has successfully restored the vision of a 13-year-old boy suffering from the rare genetic condition dystrophic epidermolysis bullosa (DEB), which scarred his eyes. The groundbreaking case, detailed in a recent publication in The…

Eyenovia announced the successful completion of the FDA inspection for its facility in Redwood City, California, marking a significant milestone in the company’s manufacturing capabilities. This achievement signals the green light for the facility to engage in final assembly, packaging,…

Ophthalmologists at New York Eye and Ear Infirmary of Mount Sinai (NYEE) have pioneered a novel protocol aimed at swiftly diagnosing eye stroke and expediting care to prevent irreversible vision loss. Published in Ophthalmology, their study outlines the use of…

In a move signaling a strategic expansion, Mainline Instruments and Oculus Optikgeräte have announced the extension of their successful partnership into the ophthalmology sector for the UK and Ireland markets. This collaboration aims to introduce cutting-edge ophthalmic solutions, catering to…

In 2023, the ophthalmic industry witnessed a transformative landscape shaped by strategic company acquisitions, setting the stage for significant advancements and collaborations. The top 10 acquisitions of the year highlighted a dynamic shift in the competitive dynamics of the sector.…

Meridian Medical, headquartered in Switzerland, announced the launch of its line of ophthalmic lasers in the United States, following a series of FDA approvals. The FDA approval, received on January 25, includes the MR Q family of lasers, which offer…

Okyo Pharma’s first investigational new drug (IND) application for OK-101 has been cleared by the U.S. Food and Drug Administration (FDA), marking a pivotal moment in the development of potential therapies for neuropathic corneal pain. Initially proposed as an open-label…

The European Society of Cataract and Refractive Surgeons (ESCRS) Winter Meeting in Frankfurt at Congress Centre Messe Frankfurt, scheduled for February 15-18, 2024, promises an array of new sessions and activities aimed at fostering collaboration, learning, and networking within the…

Glaukos has received FDA approval for iDose TR (travoprost intracameral implant 75 mcg), a groundbreaking prostaglandin analog intracameral implant designed to reduce intraocular pressure (IOP) in individuals with ocular hypertension or open-angle glaucoma. The iDose TR procedure, sanctioned for a…

Eyenovia Inc. announced that the FDA has approved Mydcombi, a groundbreaking ophthalmic spray that induces mydriasis for diagnostic procedures and short-term pupil dilation. This marks the first approved, fixed-dose combination of tropicamide and phenylephrine in the United States and also…