
Newsletter Subscribe
Enter your email address below and subscribe to our newsletter


Enter your email address below and subscribe to our newsletter

(Image Credit: AdobeStock/Tada Images) The US Food and Drug Administration (FDA) has granted Fast Track Designation to Complement Therapeutics’ CTx001, the company’s lead gene therapy candidate for the treatment of geographic atrophy (GA) secondary to age-related macular degeneration (AMD). The…

(Image Credit: AdobeStock/Timon) Nanoscope has received a new patent (US Patent No. 12459977) from the US Patent and Trademark Office (USPTO) for the company’s Multi-Characteristic Opsin (MCO) technology platform as a novel and innovative synthetic opsin for optogenetic modulation. MCO is…

(Image Credit: AdobeStock/Ilya) SpliceBio has dosed the first patient in the Part B dose-expansion portion of its phase 1/2 ASTRA clinical trial (NCT06942572) evaluating SB-007 for the treatment of Stargardt disease. SB-007 is an investigational dual adeno-associated virus (AAV) gene…

Glaucoma care is evolving through the integration of artificial intelligence, advanced procedures, and personalized treatment strategies. (Image credit: AdobeStock/thodonal) As the ophthalmology community observes Glaucoma Awareness Month this January, glaucoma specialists point to incremental integration of technologies that support earlier…

(Image Credit: Adobe Stock/fotofabrika) Ollin Biosciences has announced positive topline results from its randomized, head-to-head phase 1b JADE clinical study comparing OLN324 to faricimab (Vabysmo). The JADE clinical study enrolled more than 160 patients with diabetic macular edema (DME) or…

Outlook Therapeutics, Inc. reported that the FDA has issued a complete response letter (CRL) concerning the resubmitted biologics license application (BLA) for ONS-5010/LYTENAVA (bevacizumab-vikg).1,2 The letter noted that the agency cannot approve the application in its current form for the…

Incorporating data from low- and middle-income countries may help redefine how ophthalmology trials are designed, evaluated, and applied in practice (Image credit: AdobeStock/PUTRABATARA) Innovation in ophthalmology is often measured by what reaches the clinic—but the path to those breakthroughs begins…

In this segment, the panel compares the pivotal QUASAR trial of aflibercept 8 mg in retinal vein occlusion (RVO) with the BALATON and COMINO trials evaluating faricimab. They note that all three studies demonstrated robust visual and anatomic improvements—typically gains…

Editor’s Note: This content was generated with the assistance of AI. From first-in-class pharmacologic therapies to next-generation retinal durability strategies and less invasive surgical platforms, 2025 emerged as a pivotal year for the ophthalmic pipeline. Over the past 12 months,…
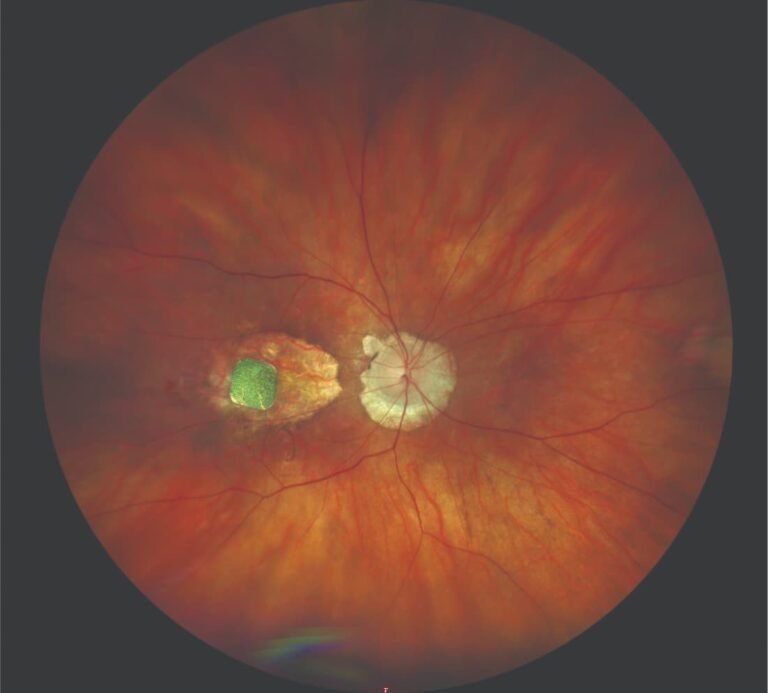
The implant is shown placed subretinally in an eye with geographic atrophy due to age-related macular degeneration. (Image courtesy of Frank G. Holz, MD, FEBO, FARVO) (PRIMA) system implant restored central vision and led to a significant improvement in visual…