Patient Education
Share your love

Lighting and Activity in Elderly with Vision Impairment
A recent study published online in JAMA Ophthalmology sheds light on the significant impact of home environment, particularly lighting, on the activity levels of older adults with visual impairment. Conducted by Seema Banerjee, Ph.D., and colleagues from Johns Hopkins University,…

2023 Recap: Top 10 Acquisitions in the Ophthalmic Industry
In 2023, the ophthalmic industry witnessed a transformative landscape shaped by strategic company acquisitions, setting the stage for significant advancements and collaborations. The top 10 acquisitions of the year highlighted a dynamic shift in the competitive dynamics of the sector.…

Meridian Medical Introduces Ophthalmic Laser Line in US
Meridian Medical, headquartered in Switzerland, announced the launch of its line of ophthalmic lasers in the United States, following a series of FDA approvals. The FDA approval, received on January 25, includes the MR Q family of lasers, which offer…

Nicox and Kowa Strike Deal for NCX 470 in Japan
Nicox SA announced an agreement with Kowa, granting the latter exclusive rights in Japan for the development and commercialization of NCX 470, a promising nitric oxide (NO)-donating bimatoprost eye drop designed for reducing intraocular pressure (IOP) in individuals with glaucoma…

FDA Gives Green Light to OK-101 Trial for NCP Treatment
Okyo Pharma’s first investigational new drug (IND) application for OK-101 has been cleared by the U.S. Food and Drug Administration (FDA), marking a pivotal moment in the development of potential therapies for neuropathic corneal pain. Initially proposed as an open-label…

New Features at ESCRS Winter Meeting in Frankfurt
The European Society of Cataract and Refractive Surgeons (ESCRS) Winter Meeting in Frankfurt at Congress Centre Messe Frankfurt, scheduled for February 15-18, 2024, promises an array of new sessions and activities aimed at fostering collaboration, learning, and networking within the…

Low-Dose Atropine Eye Drops & Myopia Progression
According to the National Eye Institute (NEI), a trial conducted by the Pediatric Eye Disease Investigator Group (PEDIG) and funded by the NEI revealed that the use of low-dose atropine eye drops (concentration 0.01%) did not demonstrate superiority over a…

Glaukos Secures FDA Approval for iDose TR
Glaukos has received FDA approval for iDose TR (travoprost intracameral implant 75 mcg), a groundbreaking prostaglandin analog intracameral implant designed to reduce intraocular pressure (IOP) in individuals with ocular hypertension or open-angle glaucoma. The iDose TR procedure, sanctioned for a…

Eyenovia’s Mydcombi Approved by the FDA
Eyenovia Inc. announced that the FDA has approved Mydcombi, a groundbreaking ophthalmic spray that induces mydriasis for diagnostic procedures and short-term pupil dilation. This marks the first approved, fixed-dose combination of tropicamide and phenylephrine in the United States and also…
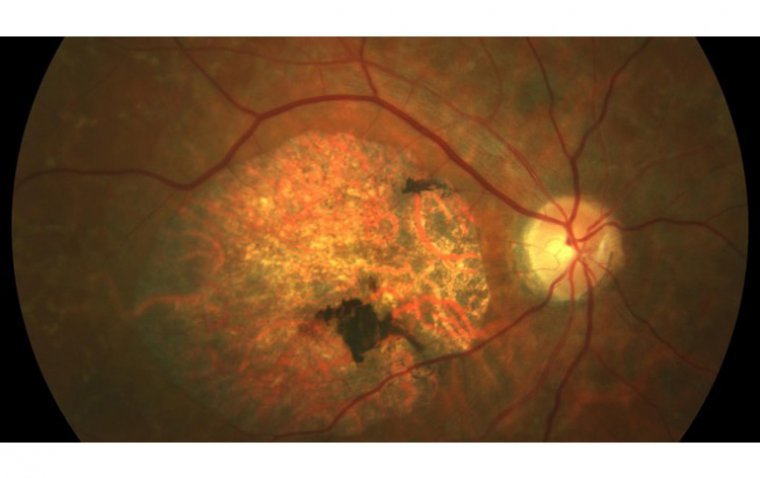
What Is Stargardt Disease? | OBN
What Is Stargardt Disease? Stargardt disease, also known as Stargardt macular degeneration or juvenile macular degeneration, is an inherited eye disease that affects the macula, which is the central part of the retina responsible for sharp, central vision. Stargardt disease…





