
Newsletter Subscribe
Enter your email address below and subscribe to our newsletter


Enter your email address below and subscribe to our newsletter

Clearside Biomedical announced it has entered into an agreement with HealthCare Royalty Partners in a deal worth up to $65 million. The money received from the deal will be put to use by Clearside to fund the ongoing clinical testing…
Noninferiority randomised trials in ophthalmology – Eye Nature Source: Author: | Date: 2023-03-03 08:00:00 Source: Author: | Date: 2023-03-03 08:00:00

Topcon Healthcare Solutions and OphtAI, a joint venture specialized in Artificial Intelligence (AI) for ophthalmology, have announced a partnership to integrate OphtAI into Topcon’s Harmony Referral System, and provide eye care professionals with smarter access to AI services. OphtAI’s state-of-the-art…
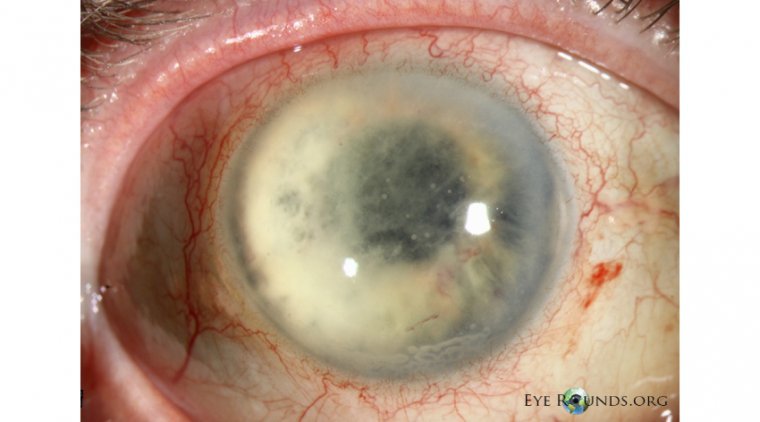
South Korean ophthalmologists reported a fungal endophthalmitis outbreak after cataract surgery due to contaminated viscoelastic material. Researchers from the Department of Ophthalmology at Korea University Guro Hospital in Seoul, Korea, under the direction of Seong Woo Kim, MD, PhD, found…

Johnson & Johnson Vision has announced that they have received CE Mark approval for the ELITA Femtosecond Laser System. The laser is designed to perform refractive correction on patients with myopia, with or without astigmatism, using the SILK (Smooth Incision…

According to a recent systematic review and meta-analysis published online on Feb. 16 in JAMA Ophthalmology, the Amsler grid has a 67 percent sensitivity for detecting neovascular age-related macular degeneration (AMD) when compared with healthy controls. In a systematic literature…
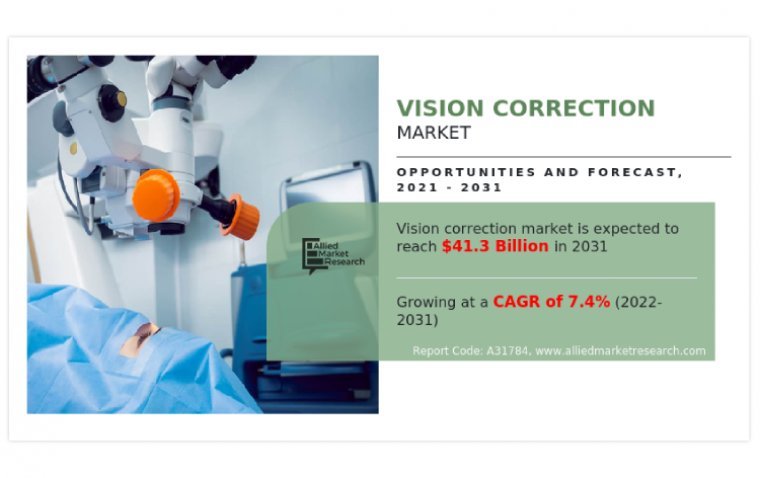
According to recent market research by Allied Market Research, the global vision correction market was worth $20,302.8 million in 2021, and is expected to grow at a compound annual growth rate (CAGR) of 7.4% from 2022 to 2031, reaching $41,252.7…

The European Parliament voted in favor of delaying the implementation of the new Medical Device Regulation (MDR) and approved the European Commission’s plan, which was adopted in January. The plan involves extending the deadlines for compliance with the new certification…

Frontera Therapeutics has initiated a clinical trial for its gene therapy, FT-002, which is intended for the treatment of X-linked retinitis pigmentosa (XLRP). The first patient has already been given a dose of the gene therapy product. FT-002 is Frontera’s…

The FDA issued a Warning Letter1 on January 31, 2023, to RightEye, LLC, the manufacturer of the RightEye Vision System, for misbranding and adulteration. The RightEye Vision System is a Class II Nystagmograph medical device that has been cleared “recording,…