Patient Education
Share your love

EC Approves Opuviz, Eylea Biosimilar
Samsung Bioepis and Biogen have announced that the European Commission (EC) has approved Opuviz 40 mg/mL for injection. This biosimilar, also known as SB15, references the widely used treatment Eylea (aflibercept). The approval encompasses a range of retinal conditions, providing…

FDA Reviews Aldeyra’s Dry Eye Drug
Aldeyra Therapeutics has announced a major milestone as the FDA formally accepted its resubmitted New Drug Application (NDA) for reproxalap, a topical ocular therapy for the treatment of dry eye disease. Alongside this regulatory progress, Aldeyra has expanded its exclusive…

SERI and ZEISS Partner to Improve Vision Care
The Singapore Eye Research Institute (SERI) and Carl Zeiss Meditec have launched a groundbreaking collaboration aimed at advancing cataract and refractive surgery outcomes. The initiative, called Ophthalmic Tech Innovation by ZEISS and SERI (OPTIZS), involves a combined investment of nearly…

Alcon’s IOL Faces Probe by CDSCO
The Central Drugs Standard Control Organisation (CDSCO) has formed a committee to investigate allegations of quality issues with an intraocular lens (IOL) supplied by Alcon. The Swiss-American company specializes in eye care products, including lenses for cataract surgery. Specifics of…

OBN Spotlight TV: Interview with Hillary A. Golden
In late October, Medicare introduced proposed Local Coverage Determinations (LCDs) that aimed to impose restrictions on Minimally Invasive Glaucoma Surgery (MIGS) procedures. These LCDs were met with significant concern and opposition from medical professionals, patient advocacy groups, and the ophthalmology…
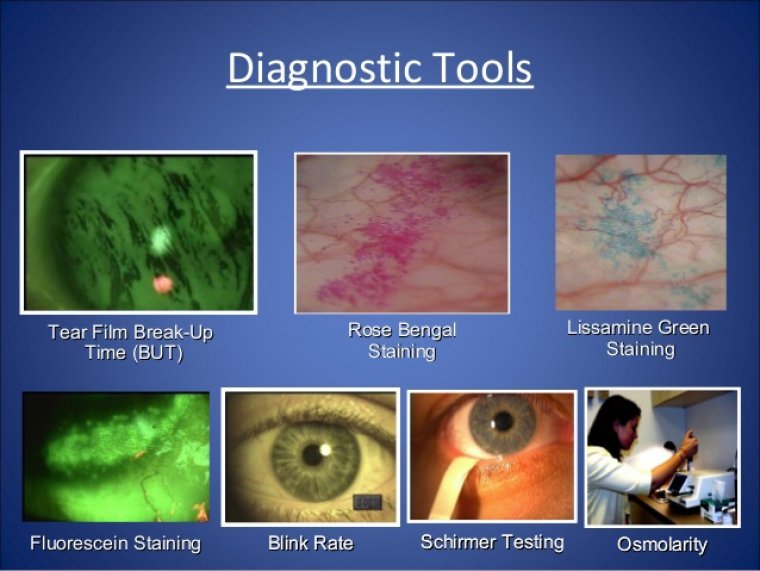
Innovations in Dry Eye Testing: New Diagnostic Tools
Dry eye disease (DED), also known as keratoconjunctivitis sicca, is one of the most common ophthalmic conditions, affecting hundreds of millions of people worldwide. Recent technological advances and research targeting DED have led to the emergence of new definitions and…

AbbVie Introduces Refresh Tears PF and Refresh Relieva PF Xtra
AbbVie has introduced two new preservative-free (PF) lubricant eye drop products─Refresh Tears PF and Refresh Relieva PF Xtra. These new additions aim to enhance the options available for eye care with innovative features tailored for user convenience and eye health.…

Eyenovia Pediatric Myopia Trial Falls Short
Eyenovia has announced that its Phase 3 CHAPERONE study, aimed at treating pediatric progressive myopia, failed to meet its primary endpoint. The trial evaluated the company’s proprietary drug-device combination using low-dose atropine delivered via the Optejet platform. A review by…

Cequa Shows Positive Phase 4 Results for Dry Eye
Sun Pharmaceutical has shared new Phase 4 clinical trial data, showcasing the efficacy of Cequa (cyclosporine ophthalmic solution) 0.09% in treating dry eye disease (DED). The results reveal that Cequa provides sustained improvements in patients whose symptoms were inadequately managed…

Bausch + Lomb Recycles 94M Eye Care Waste Units
Bausch + Lomb has announced a major milestone in its sustainability efforts. Through its exclusive recycling initiatives—ONE by ONE Recycling and Biotrue Eye Care Recycling programs—the company has collected 94,119,275 units of used contact lenses, eye care products, and lens…





