Patient Education
Share your love

Okyo Pharma’s Phase 2 Trial for Neuropathic Corneal Pain
Okyo Pharma announced its plan to initiate a phase 2 clinical trial for OK-101 in the treatment of neuropathic corneal pain (NCP), scheduled to begin in the third quarter of 2024. This one-year study follows promising results from preclinical animal…

Opthea Forms Medical Advisory Board with Global Retina Experts
Opthea announced the formation of its Medical Advisory Board (MAB), comprised of ten distinguished thought leaders in the field of retina from across the globe. The board includes experts from the United States, Argentina, Australia, China, France, Germany, and Israel,…
Screen Use for Kids – American Academy of Ophthalmology
Screen Use for Kids American Academy of Ophthalmology Source: Author: | Date: 2024-07-19 07:00:00 Source: Author: | Date: 2024-07-19 07:00:00

NexThera Submits IND Application for NT-101 for Wet AMD
NexThera has officially submitted a Phase 1/2a Investigational New Drug (IND) application to the U.S. Food and Drug Administration (FDA) for NT-101, an innovative non-invasive eye drop treatment targeting wet age-related macular degeneration (wet AMD). Proprietary Delivery Platform: Advancing Treatment…

ENTOD Pharma Introduces New Eye Drop Preservative System
Mumbai-based ENTOD Pharmaceuticals has unveiled a novel advancement in ophthalmic care with the introduction of EyeBS, a new eye drop preservative system. EyeBS is specifically designed to supplant traditional detergent-based preservatives such as Benzalkonium Chloride (BAK) and Polyquaternium-1, offering a…
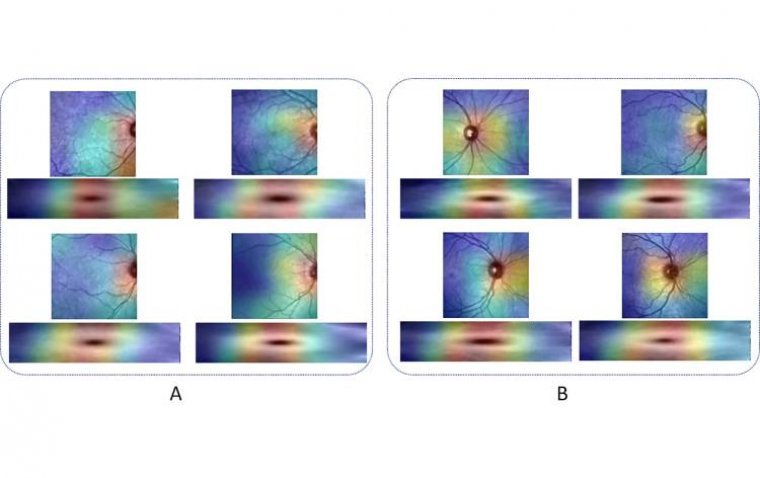
Researchers Discover New Imaging Technique for MS Diagnosis
Researchers from Durham University, UK, and Isfahan University of Medical Sciences, Iran, have discovered a pioneering method for diagnosing multiple sclerosis (MS) using advanced eye imaging techniques. Published in the journal Translational Vision Science & Technology, their work introduces a…

Funding Boost for PulseMedica’s Eye Floater Technology
PulseMedica has successfully raised a total of $12 million in their recent pre-Series A financing round, marking a significant milestone in their pursuit of innovative eye floater diagnostic and treatment technologies. Accelerating Clinical Validation and Expansion Efforts This funding round…
Ophthalmologic Manifestations of Acute Leukemia Are Detectable
Acute leukemia can present with various ophthalmologic manifestations, which are detectable at both initial presentation and relapse, according to a study published online on July 10 in the Annals of Hematology. Dr. Dina N. Laimon and her colleagues from Mansoura…
Study Highlights Significant Drug Waste from SUCD for Eye Drops
A recent report published online in Ophthalmology reveals alarming levels of drug wastage and associated costs due to self-imposed use cessation dates (SUCD) for multiuse eye drop bottles. John M. Tan and colleagues from the Icahn School of Medicine at…
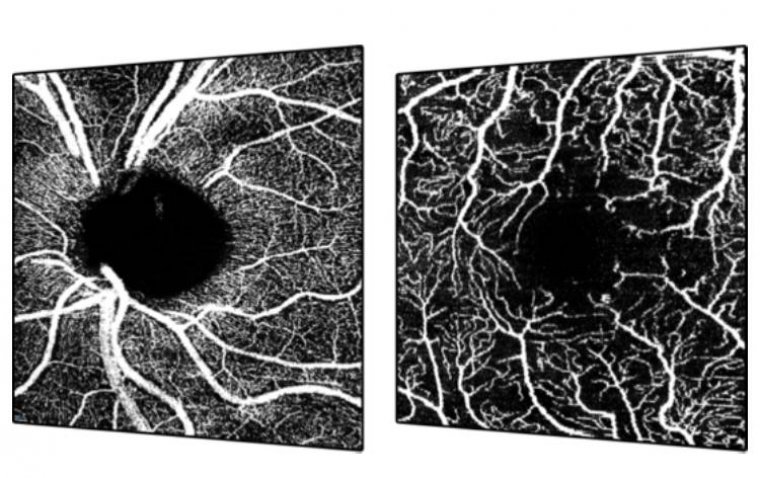
FDA Clears Enhanced SPECTRALIS® OCTA Module by Heidelberg
Heidelberg Engineering announced the FDA clearance of its upgraded SPECTRALIS® OCTA Module featuring SHIFT technology. This advancement promises to significantly reduce acquisition time by 50%, marking a pivotal development in clinical efficiency and diagnostic precision within ophthalmology. Enhancing Clinical Utility…





