
Newsletter Subscribe
Enter your email address below and subscribe to our newsletter


Enter your email address below and subscribe to our newsletter
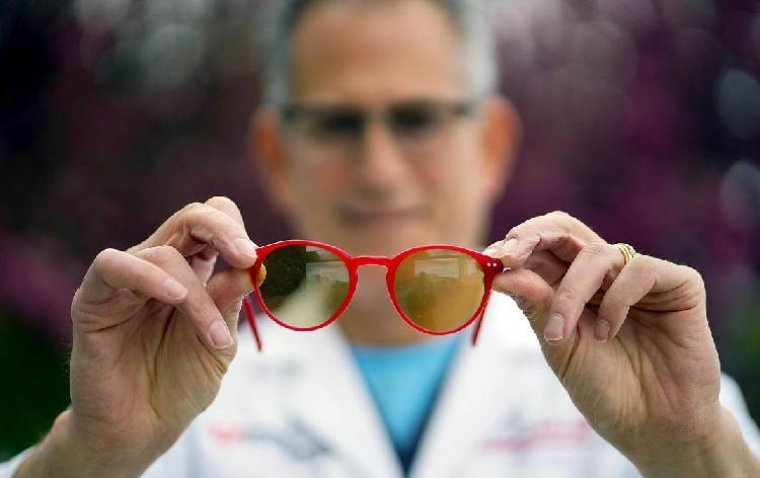
This spring, individuals grappling with migraines find relieve with a novel solution for managing light sensitivity, thanks to new eyeglass lens technology developed by a physician-scientist at the University of Utah John A. Moran Eye Center. The Impact of Migraine…
Fairer AI in ophthalmology via implicit fairness learning for mitigating sexism and ageism Nature Source: Author: | Date: 2024-06-04 07:00:00 Source: Author: | Date: 2024-06-04 07:00:00

LambdaVision, a pioneering company dedicated to developing a protein-based artificial retina aimed at restoring sight lost to retinal degenerative diseases, announced the successful first closing of its seed round to advance its artificial retina development. The funding injection will fuel…

Sightpath Medical, a leading provider of mobile and fixed ophthalmic surgical services, has recently completed the acquisition of PennVista, a renowned LASIK services business. PennVista specializes in offering cutting-edge equipment and skilled technicians to surgery centers and hospitals across Pennsylvania,…
Lessons from a career in ophthalmology Emory School of Medicine Source: Author: | Date: 2024-06-03 07:00:00 Source: Author: | Date: 2024-06-03 07:00:00

Quantel Medical, a division of Lumibird Medical, has unveiled its latest innovation with the launch of the POCKET III handheld pachymeter. This compact device, according to Quantel, sets a new standard as the lightest connected handheld pachymeter on the market,…
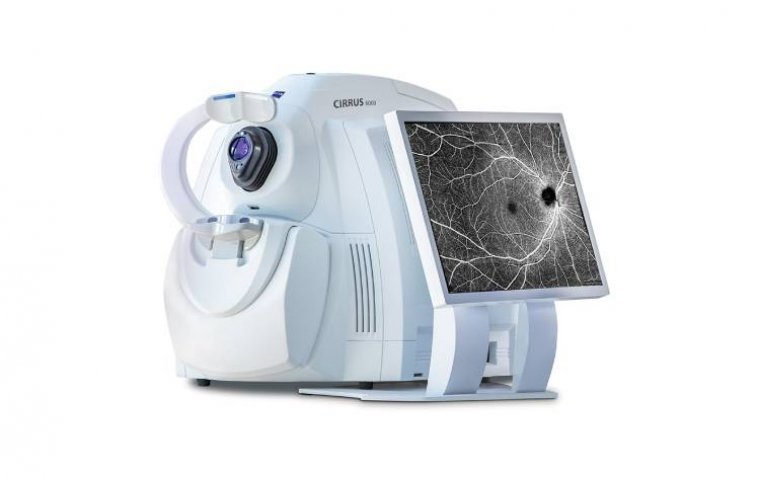
Zeiss Medical Technology has introduced a series of enhancements to the Cirrus 6000, promising a more streamlined and data-centric workflow for ophthalmologists. This update boasts the largest OCT reference database in the US market and fortified cybersecurity features. With recent…

The U.S. Food and Drug Administration (FDA) has qualified the Assessment of IntraOcular Lens Implant Symptoms (AIOLIS), developed by the American Academy of Ophthalmology (AAO), as a Medical Device Development Tool (MDDT). This designation empowers medical device sponsors to employ…

The increasing prevalence of digital devices in children’s lives has sparked a growing concern among parents, educators, and healthcare professionals about the potential impacts on eye health. As screens become an integral part of education, entertainment, and social interaction, understanding…

In a significant development for the medical community, Drug Farm announced that the U.S. Food and Drug Administration (FDA) has granted clearance for the Investigational New Drug (IND) application of DF-003. This novel drug, hailed as a first-in-class oral medication,…