
Newsletter Subscribe
Enter your email address below and subscribe to our newsletter


Enter your email address below and subscribe to our newsletter

FCI Ophthalmics announced the launch of EzyPor in the United States. EzyPor is a polyethylene orbital implant with a proprietary suturing platform that is suggested for enucleation, evisceration, and secondary implantation. Ezypor is developed with a smooth anterior suturing surface…

HOYA Surgical Optics (“HSO”), the intraocular lens and related products division of HOYA Group, today announced that it has completed the acquisition of medical device companies Mid Labs and Fritz Ruck. Mid Labs is a California-based company focused on the…

Development of active glasses for mitigating presbyopia has become an active area of research, facilitated by the miniaturization of focus-tunable lenses as well as the increased processing power of modern computers. Alcon the global leader in eye care dedicated to…

The global ophthalmic sutures market size is expected to reach USD 532.8 million by 2026, expanding at a CAGR of 6.4% over the forecast period, according to a new report by Grand View research, Inc. Key factors driving the market…

Cataract surgery is the most frequently performed operative procedure worldwide, typically concludes with the implantation of an artificial intraocular lens (IOL) to correct aphakia (absence of the crystalline lens). The surgery—which involves removing the eye’s clouded lens and replacing it…
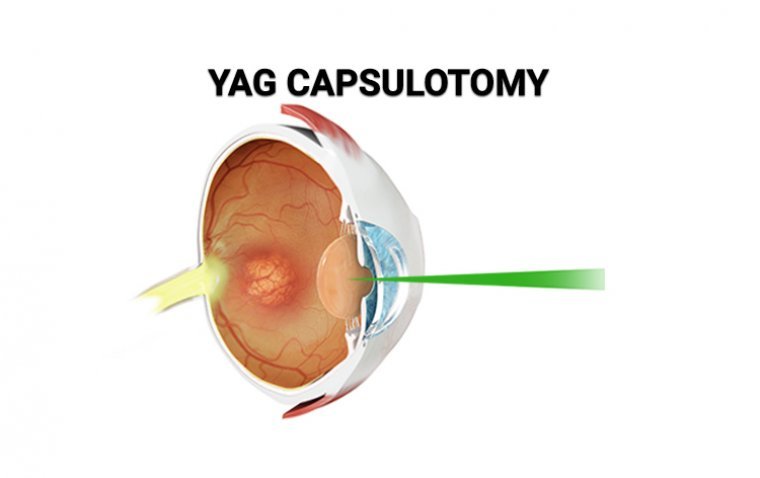
Cataract surgery has revolutionized the way we restore vision, but what happens when clarity begins to fade post-surgery? This is where YAG laser capsulotomy steps in, offering a solution that restores vision and rejuvenates the eyes. In this article, we’ll…

Lupin Limited, a global pharmaceutical company, has officially received approval from the US FDA for its Abbreviated New Drug Application for Bromfenac Ophthalmic Solution, 0.09%. This approval allows Lupin to introduce a generic counterpart to Bausch & Lomb Inc.’s Bromday…
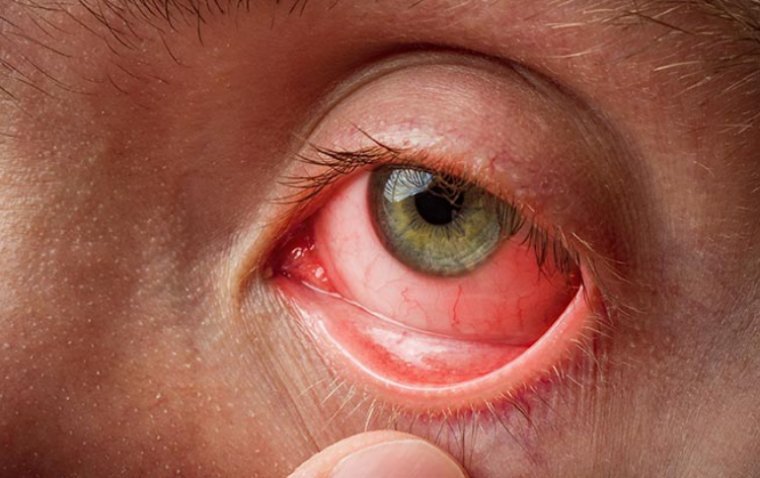
Conjunctivitis, commonly known as “pink eye,” is a prevalent eye condition that affects people of all ages. Characterized by redness, itching, and discomfort in the eyes, conjunctivitis can be caused by various factors, including viruses, bacteria, allergies, and irritants. Understanding…
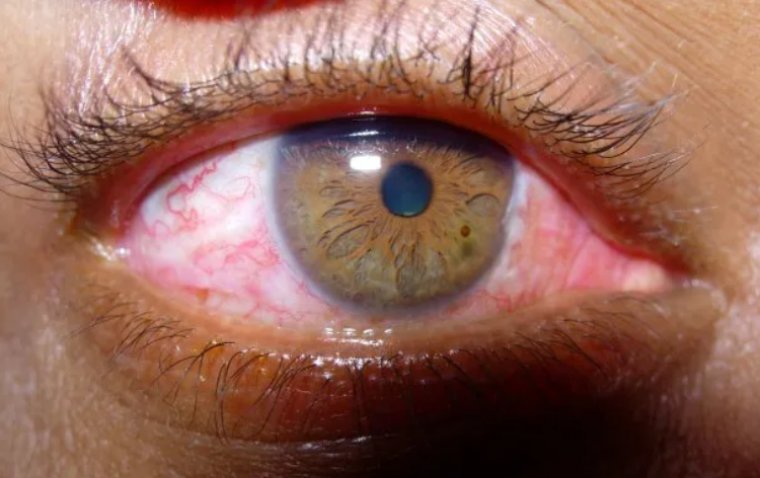
Conjunctival injection, commonly known as red eyes, is a condition characterized by the appearance of bloodshot or pinkish eyes. It occurs when the blood vessels on the conjunctiva, the thin membrane covering the front surface of the eye, become dilated…

The Medicines and Healthcare products Regulatory Agency (MHRA) has recently issued a National Patient Safety Alert due to reports of increased intraocular pressure in patients recently implanted with EyeCee One preloaded and EyeCee One Crystal preloaded intraocular lenses (IOLs), which…