
Newsletter Subscribe
Enter your email address below and subscribe to our newsletter


Enter your email address below and subscribe to our newsletter
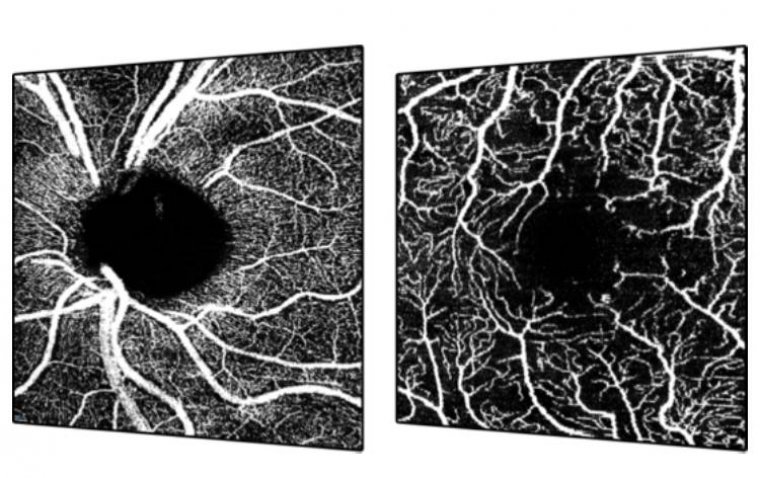
Heidelberg Engineering announced the FDA clearance of its upgraded SPECTRALIS® OCTA Module featuring SHIFT technology. This advancement promises to significantly reduce acquisition time by 50%, marking a pivotal development in clinical efficiency and diagnostic precision within ophthalmology. Enhancing Clinical Utility…

Genentech announced the reintroduction of Susvimo (ranibizumab injection) 100 mg/mL for intravitreal use via ocular implant, aimed at treating wet age-related macular degeneration (AMD). This decision follows the FDA’s approval of a post-approval supplement to the Biologics License Application, which…

Outlook Therapeutics announced that the UK Medicines and Healthcare products Regulatory Agency (MHRA) has granted marketing authorization for Lytenava (bevacizumab gamma) for the treatment of wet age-related macular degeneration (AMD) in the UK. Lytenava is the first and only authorized…

Glaukos Corporation has confirmed the activation of the CMS Healthcare Common Procedure Coding System’s J-code—J7355—for its iDose TR product. This development marks a significant milestone in enhancing patient access and streamlining coverage and payment processes for the innovative treatment, according…

Formycon AG and its licensing partner Klinge Biopharma GmbH announced the U.S. Food and Drug Administration’s (FDA) approval of FYB203/AHZANTIVE®, a biosimilar to Eylea®. FYB203/AHZANTIVE® has been approved by the FDA for the treatment of patients with Age-Related Neovascular (wet)…
Beacon Therapeutics has successfully raised $170 million in Series B funding, marking a significant step forward in advancing its portfolio of ophthalmic gene therapies. The funds are earmarked to support the ongoing clinical development of Beacon’s lead candidate, AGTC-501, designed…
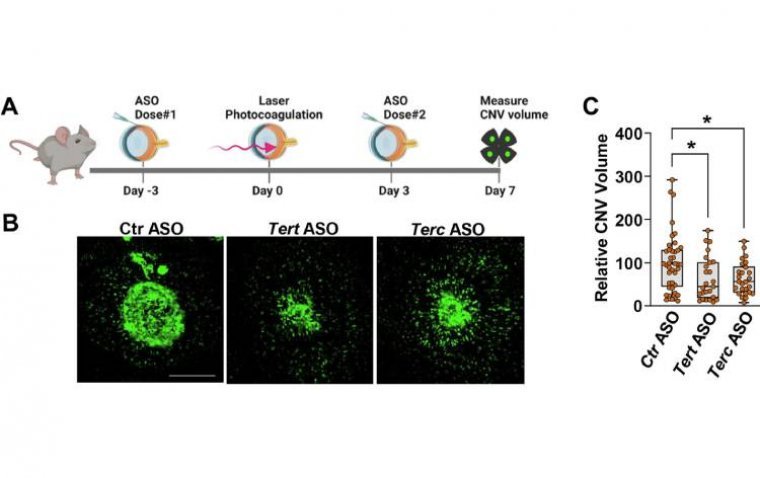
A recent study conducted on mice has provided promising insights into a potential alternative treatment for the “wet” version of age-related macular degeneration (AMD). Researchers identified an enzyme, telomerase, which plays a critical role in the abnormal blood vessel growth…

Alcon has finalized its acquisition of BELKIN Vision, marking a significant expansion of its glaucoma treatment offerings. The acquisition, valued at $81M upfront, with potential additional payments up to $385M contingent on sales milestones, includes BELKIN Vision’s innovative Direct Selective…
Roche announced that the Committee for Medicinal Products for Human Use (CHMP) of the European Medicines Agency (EMA) has issued a positive opinion recommending the extension of marketing authorization for Vabysmo (faricimab). This recommendation includes the treatment of visual impairment…
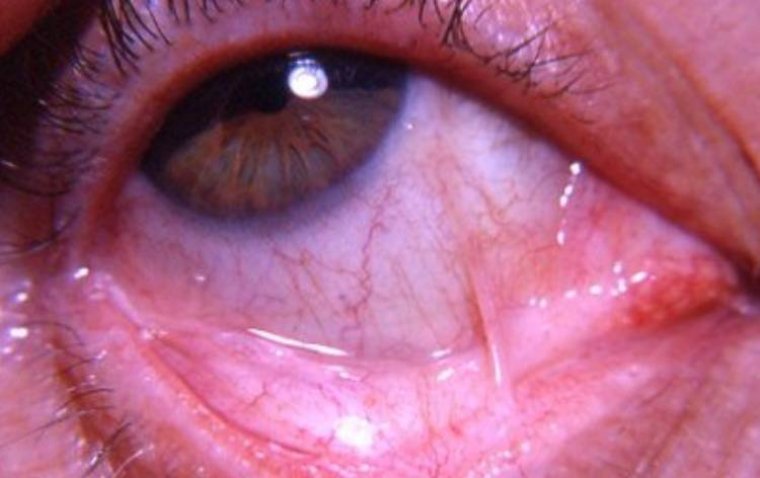
What Is Symblepharon? Symblepharon is a medical condition characterized by the adhesion of the conjunctiva, the clear membrane that covers the white part of the eye and the inside of the eyelids, to the cornea, the clear front part of…