
Newsletter Subscribe
Enter your email address below and subscribe to our newsletter


Enter your email address below and subscribe to our newsletter

The U.S. Food and Drug Administration (FDA) has qualified the Assessment of IntraOcular Lens Implant Symptoms (AIOLIS), developed by the American Academy of Ophthalmology (AAO), as a Medical Device Development Tool (MDDT). This designation empowers medical device sponsors to employ…

In a significant development for the medical community, Drug Farm announced that the U.S. Food and Drug Administration (FDA) has granted clearance for the Investigational New Drug (IND) application of DF-003. This novel drug, hailed as a first-in-class oral medication,…

Outlook Therapeutics has achieved a significant milestone with the European Commission granting Marketing Authorization for Lytenava (bevacizumab gamma), an innovative ophthalmic formulation designed for the treatment of wet AMD (Age-related Macular Degeneration). This approval marks a pivotal moment as Lytenava…

Opthea Limited announced the completion of enrollment in both the COAST and ShORe trials, constituting its pivotal Phase 3 clinical program. This program is designed to assess the safety and superior efficacy of sozinibercept in combination with standard-of-care anti-VEGF-A therapies…

Exonate announced the expansion of its Board with the appointment of Dr. Rafiq Hasan as Non-Executive Director. This strategic move aims to bolster the company’s expertise in ophthalmology, particularly within the diabetic eye disease sector, as it advances its lead…

*If you are interested in becoming a part of our OBN Spotlight TV series, reach us at info@ophthalmologybreakingnews.com. Q1: As you reflect on the recent ESCRS 2023 Congress, could you share with us the key highlights and breakthroughs in cataract…
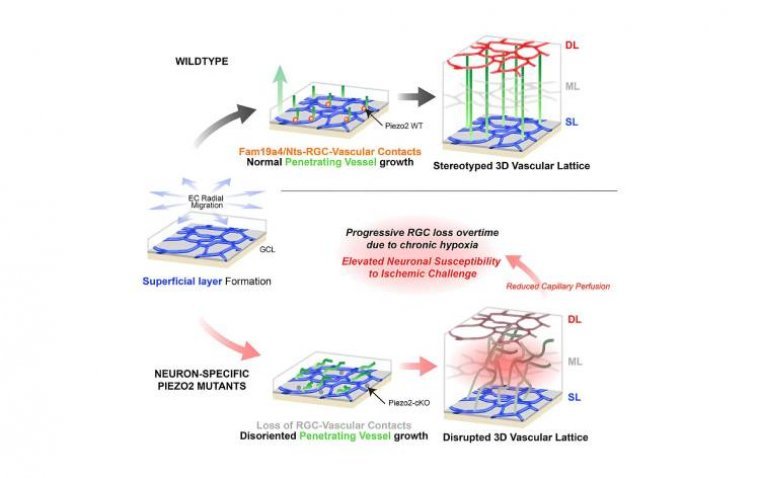
Scientists at UC San Francisco have identified a novel type of neuron that plays a crucial role in guiding the formation of the intricate lattice of blood vessels in the retina. This lattice is essential for nourishing the cells responsible…
Researchers from the National Institute for Health Research Biomedical Research Centre at Moorfields Eye Hospital and University College London Institute of Ophthalmology, London have conducted a post hoc analysis of data from two phase 3 clinical trials of pegcetacoplan (Syfovre,…

Outlook Therapeutics announced the submission of its Marketing Authorisation Application (MAA) to the United Kingdom’s Medicines and Healthcare products Regulatory Agency (MHRA) in a press release. The MAA is for bevacizumab gamma (ONS-5010/LYTENAVA), its investigational formulation of bevacizumab for the…

Rezolute announced promising topline results from its Phase 2 clinical study of RZ402. This study targeted patients with diabetic macular edema (DME) who were either naïve to or had limited exposure to anti-vascular endothelial growth factor (anti-VEGF) injections. Monumental Results…