
Newsletter Subscribe
Enter your email address below and subscribe to our newsletter


Enter your email address below and subscribe to our newsletter

The US Food and Drug Administration (FDA) has approved aflibercept injection 8 mg (EYLEA HD) from Regeneron Pharmaceuticals for the treatment of patients with macular edema following retinal vein occlusion (RVO) with up to every 8-week dosing after an initial monthly…

(Image credit: ©peopleimages.com/AdobeStock) The Data Safety Monitoring Committee (DSMC) recently completed its second scheduled review of EyePoint Pharmaceuticals’ ongoing phase 3 trial evaluating DURAVYU for the treatment of wet AMD (wAMD), EyePoint announced.1 The DSMC concluded that both the LUGANO…
(Image Credit: AdobeStock/Anna) Precise Bio has successfully treated a patient with PB-001, the company’s 3D-bio-printed corneal implant, at Rambam Medical Center in Haifa, Israel. The procedure was conducted as part of the company’s ongoing phase 1 clinical trial. PB-001 is…

The Viewpoints series “Reimagining Dry Eye Management: Targeting Tear Function for Sustained Relief” convenes leading experts in ophthalmology and optometry to discuss evolving perspectives on dry eye disease (DED). Under the moderation of Kendall Donaldson, MD, MS, the panel—comprising Cecelia…

The Viewpoints series “Reimagining Dry Eye Management: Targeting Tear Function for Sustained Relief” convenes leading experts in ophthalmology and optometry to discuss evolving perspectives on dry eye disease (DED). Under the moderation of Kendall Donaldson, MD, MS, the panel—comprising Cecelia…
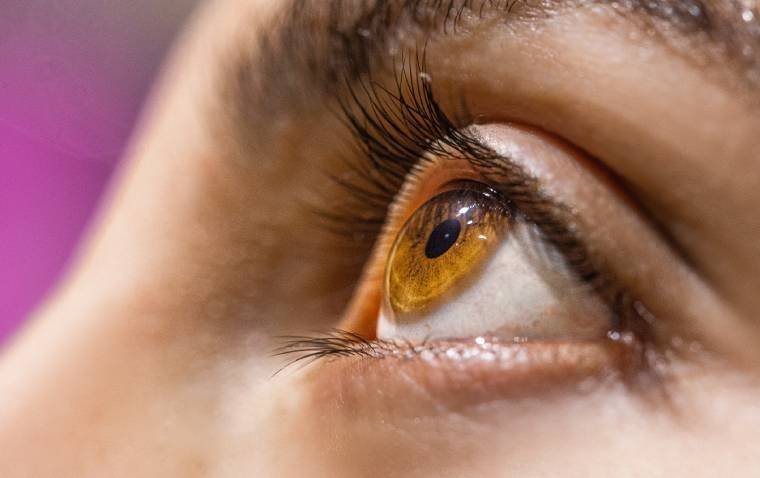
A new study from researchers at EPFL introduces a promising non-invasive brain stimulation technique aimed at restoring visual function in stroke patients with hemianopia. The treatment combines visual training with targeted neuromodulation, offering a novel approach to visual rehabilitation in…

Presbyopia continues to be one of the most significant visual limitations following cataract surgery, even with the use of advanced monofocal or extended-depth intraocular lenses (IOLs). Despite technological progress, achieving consistent vision across near, intermediate and far distance foci remains…

(Image Credit: AdobeStock/Ar_TH) Foundation Fighting Blindness has granted public access to de-identified 4-year data from individuals participating in RUSH2A, an ongoing natural history study for patients with mutations in the USH2A gene, which causes Usher syndrome type 2A or nonsyndromic retinitis pigmentosa…

(Image Credit: AdobeStock/Treecha) Opus Genetics has dosed the first patient in its OPGx-BEST1 phase 1/2 clinical trial, BIRD-1, for Best disease (BEST1). Best disease, also known as vitelliform macular dystrophy, is a rare, inherited retinal condition causing macular degeneration by…

The Enova Advanced® Extended Depth of Focus (EDoF) IOL, developed by VSY Biotechnology GmbH, represents a significant evolution in intraocular lens technology. Built on advanced optical innovation and material science, this lens is designed to offer natural vision, seamless focus, and…