
Newsletter Subscribe
Enter your email address below and subscribe to our newsletter


Enter your email address below and subscribe to our newsletter

Skye Bioscience’s phase 2a clinical trial of its SBI-100 ophthalmic emulsion in patients with primary open-angle glaucoma (POAG) or ocular hypertension (OHT) has failed to meet its primary endpoint for lowering intraocular pressure (IOP). Consequently, the company announced its decision…
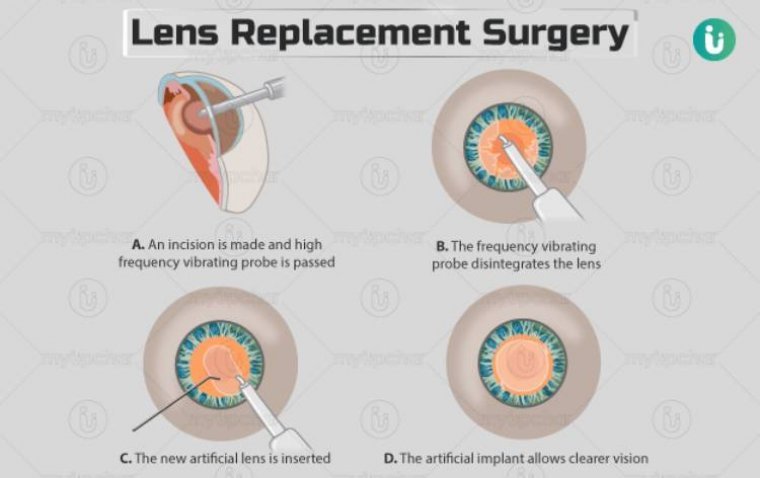
Lens replacement surgery, also known as Refractive Lens Exchange (RLE) or Clear Lens Extraction (CLE), is a procedure designed to improve vision by replacing the eye’s natural lens with an artificial one. This surgery is particularly beneficial for those who…
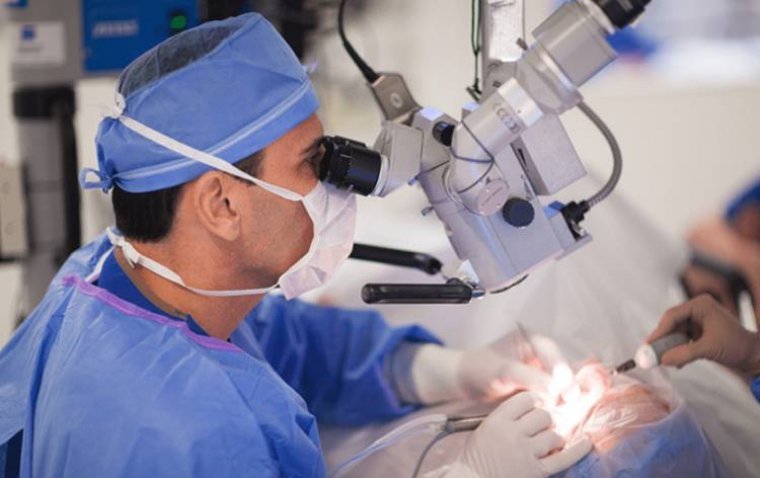
Cataract surgery is a safe, effective procedure that improves vision by removing cloudy lenses from the eyes and replacing them with artificial lenses. As technology advances and techniques improve, understanding the nuances of cataract surgery is crucial for anyone considering…

Melt Pharmaceuticals has completed the dosing of its first patient in its phase 3 program for MELT-300, marking a crucial step in evaluating the safety and efficacy of this pioneering product. MELT-300 is a non-intravenous, non-opioid tablet comprising fixed doses…

BIOPHTA, a preclinical biotech company, has successfully closed a Seed round, raising €6.5 million to advance its pioneering technology into clinical development for the treatment of glaucoma and macular edema. This significant funding injection, led by prominent European venture capital…

Sightpath Medical, a leading provider of mobile and fixed ophthalmic surgical services, has recently completed the acquisition of PennVista, a renowned LASIK services business. PennVista specializes in offering cutting-edge equipment and skilled technicians to surgery centers and hospitals across Pennsylvania,…

Quantel Medical, a division of Lumibird Medical, has unveiled its latest innovation with the launch of the POCKET III handheld pachymeter. This compact device, according to Quantel, sets a new standard as the lightest connected handheld pachymeter on the market,…
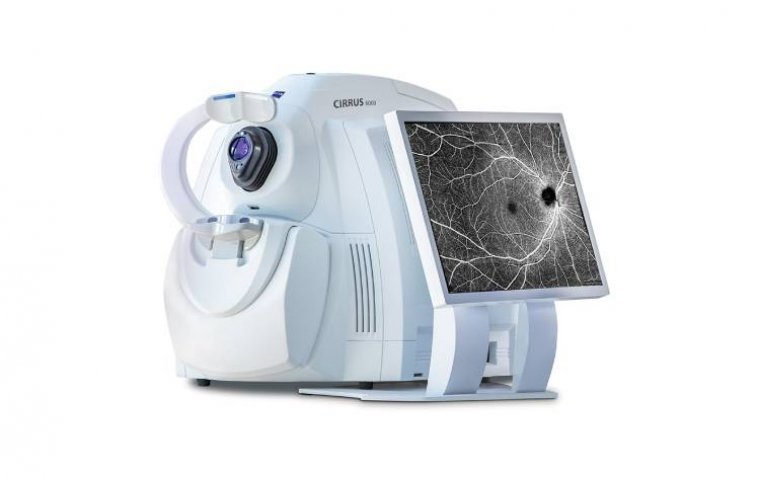
Zeiss Medical Technology has introduced a series of enhancements to the Cirrus 6000, promising a more streamlined and data-centric workflow for ophthalmologists. This update boasts the largest OCT reference database in the US market and fortified cybersecurity features. With recent…

The U.S. Food and Drug Administration (FDA) has qualified the Assessment of IntraOcular Lens Implant Symptoms (AIOLIS), developed by the American Academy of Ophthalmology (AAO), as a Medical Device Development Tool (MDDT). This designation empowers medical device sponsors to employ…

*If you are interested in becoming a part of our OBN Spotlight TV series, reach us at info@ophthalmologybreakingnews.com. Q1: As you reflect on the recent ESCRS 2023 Congress, could you share with us the key highlights and breakthroughs in cataract…