
Newsletter Subscribe
Enter your email address below and subscribe to our newsletter


Enter your email address below and subscribe to our newsletter

Photo of Alexandra Miere at the 2025 EURETINA meeting in Paris Alexandra Miere discusses retinal studies published in EURETINA journal. The first, the ACTOR study, explores vascular remodeling in neovascular AMD patients treated with ranibizumab. The second, the HERMES study,…
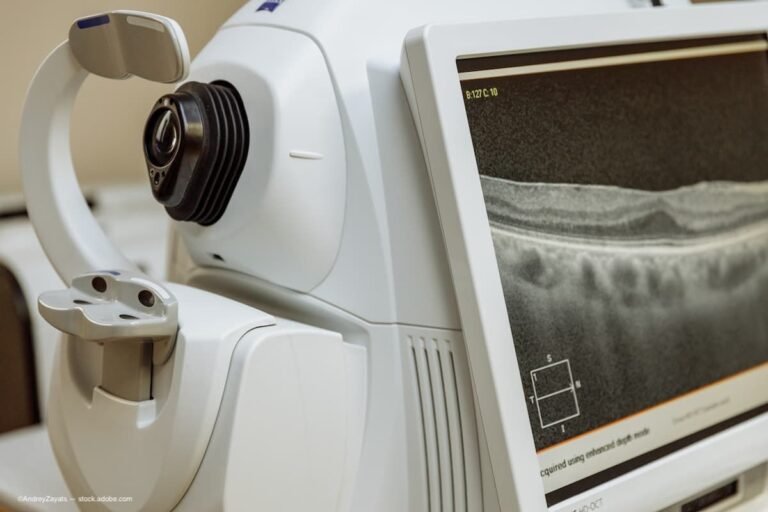
A recent conference celebrated the creation of the first prototype of the spectral optical coherence tomography (SOCT) instrument. Introduced in 2000, the prototype, which was developed at the Institute of Physics of Nicolaus Copernicus University in Toruń, Poland, was the…
\n European specialist investor GHO Capital Partners has announced the sale of its portfolio company, VISUfarma, a Netherlands-headquartered specialty pharmaceutical company focused on ophthalmology, to global pharma major Lupin. \n\n\n\n VISUfarma was created by GHO Capital in 2016 by combining…

At this year’s EyeCon meeting, Oluwatosin U. Smith, MD, of the Glaucoma Associates of Texas, reflected on her dual role—both as an educator and a leader in its development—while delivering a focused presentation on the current state of glaucoma care.…
As Ophthalmology Times celebrates its 50th anniversary, we took the opportunity to ask a thought-provoking question to some of the field’s leading experts: “In a perfect world, if you could cure one eye disease or condition with the snap of…

Photo of Boris Stanzel, MD, at the 2025 EURETINA meeting Professor Boris Stanzel, MD, presented research on methotrexate’s use in PVR retinal detachment surgery at the 2025 EURETINA congress, which was held in Paris, France. The study of 20 patients…
(Image credit: AdobeStock/phonlamaiphoto) Canadian researchers recently reported significant increases in complications during cataract surgeries performed during the COVID-19 pandemic compared with the pre-pandemic period.1 First author Nathan Lee-Wing, BS, and MD candidate at the Max Rady College of Medicine, University…

(Image Credit: AdobeStock) The US Food and Drug Administration (FDA) has approved Amneal Pharmaceuticals’ Abbreviated New Drug Application (ANDA) for bimatoprost ophthalmic solution 0.01% (2.5 mL, 5 mL and 7.5 mL).1 An ANDA is a submission to the FDA that…

(Image credit: AdobeStock/masud) The results of a new cohort study found that newly diagnosed primary open-angle glaucoma (POAG) in patients who were in the lowest wealth quartile was “substantially” less likely to reach the recommended reduction and the patients “considerably”…

(Image credit: AdobeStock/hakule) Optos and Toku have reached an agreement to make Toku’s BioAge available to users of Optos Daytona, Monaco, MonacoPro, and California devices in the United States. The companies state that by incorporating the AI solution, BioAge, eye…