
Newsletter Subscribe
Enter your email address below and subscribe to our newsletter


Enter your email address below and subscribe to our newsletter
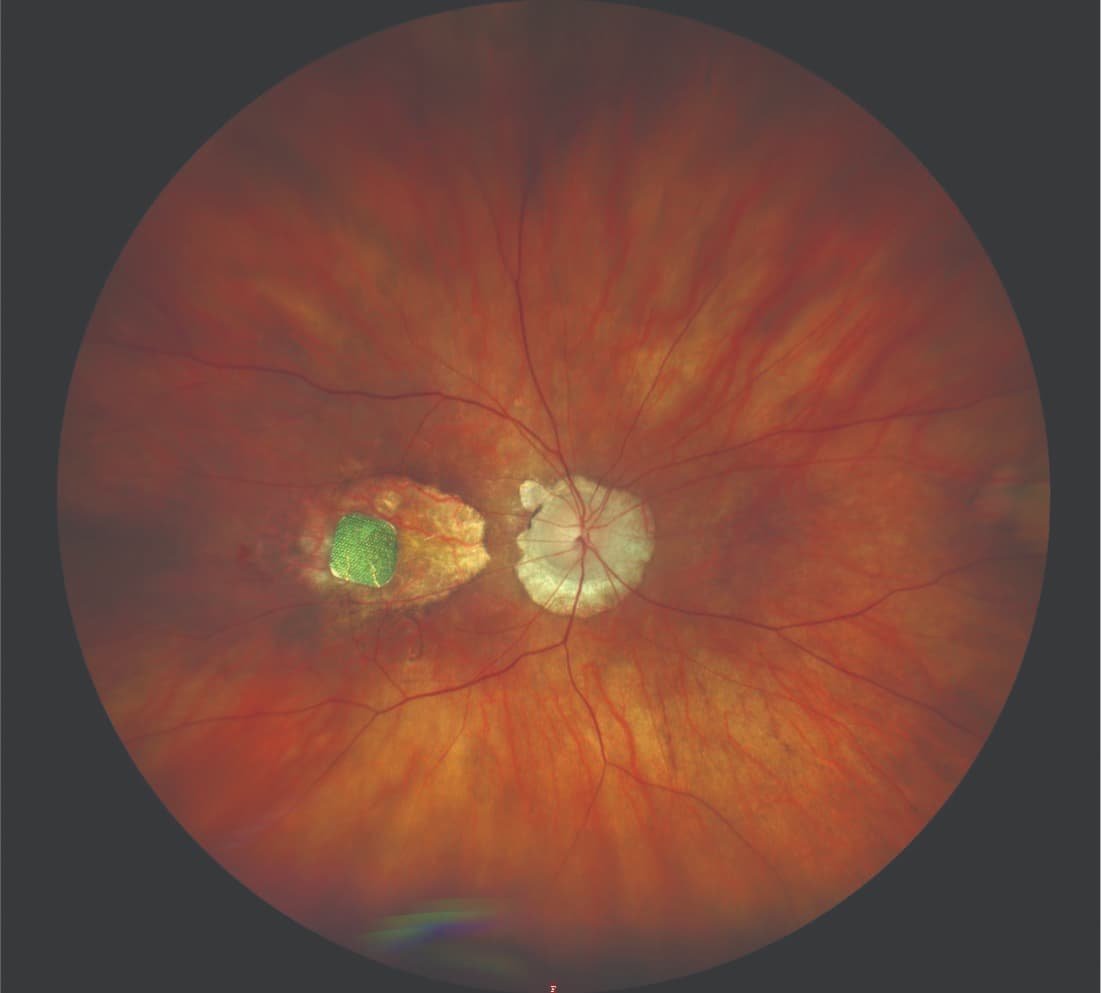
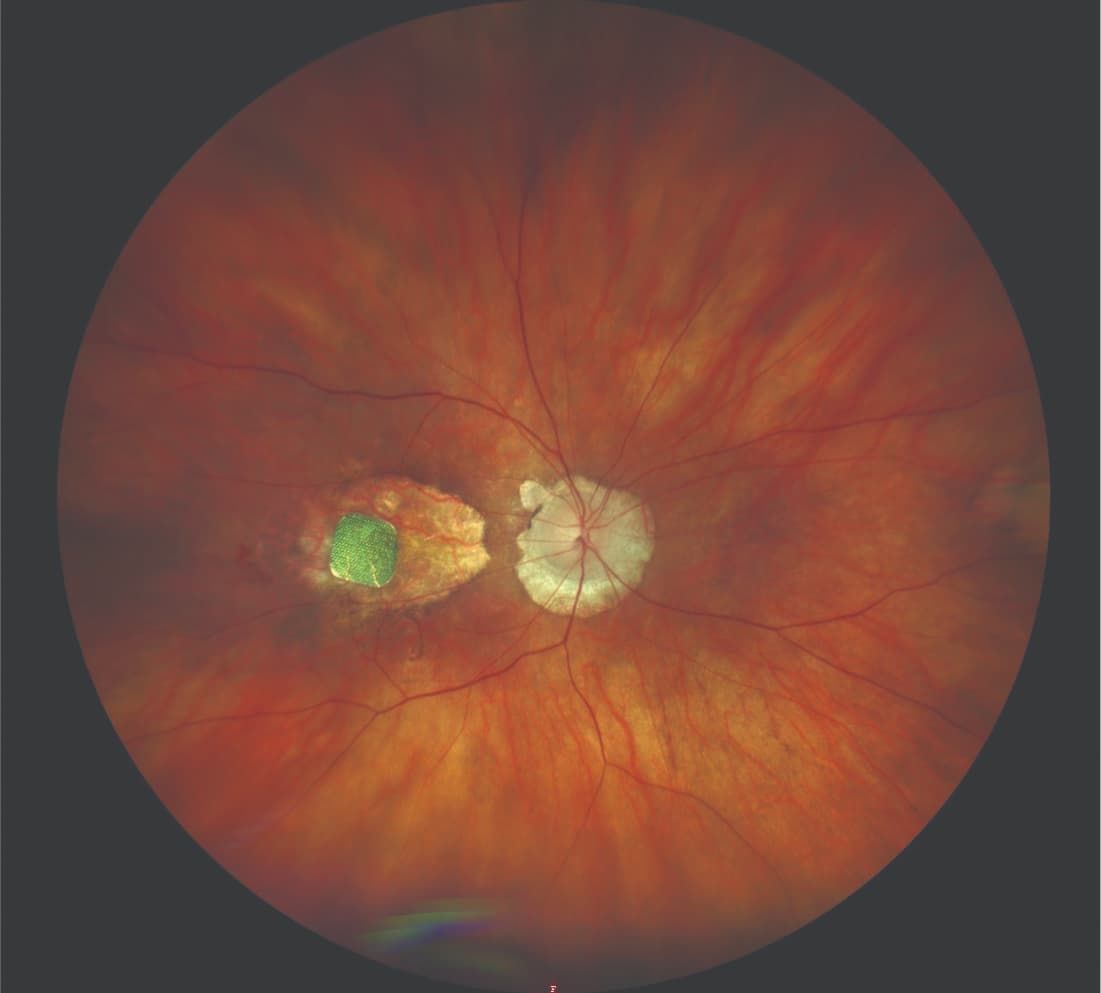
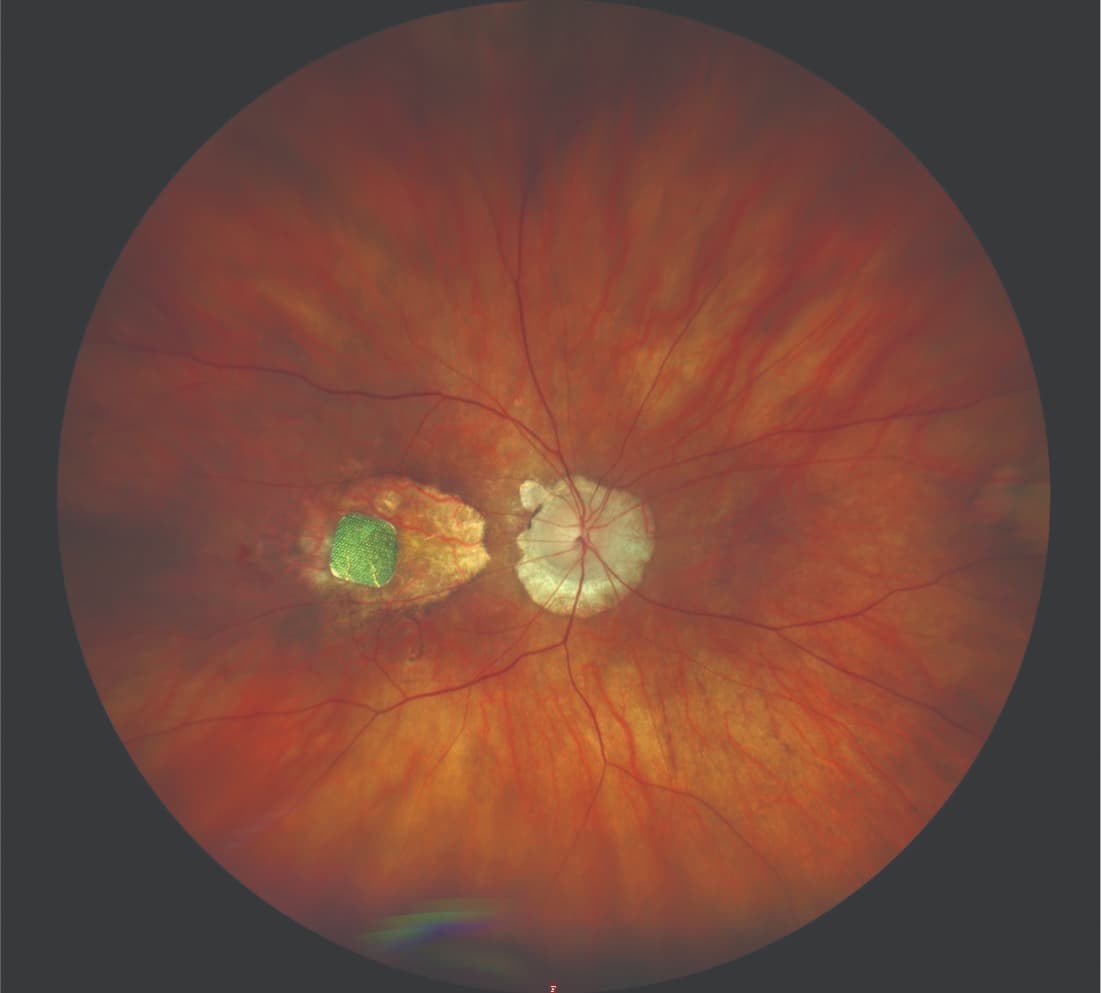
The implant is shown placed subretinally in an eye with geographic atrophy due to age-related macular degeneration. (Image courtesy of Frank G. Holz, MD, FEBO, FARVO)
(PRIMA) system implant restored central vision and led to a significant improvement in visual acuity from baseline to month 12 after implantation in patients with geographic atrophy (GA) secondary to age-related macular degeneration (AMD).1 Frank G. Holz, MD, FEBO, FARVO, and colleagues reported their results in the New England Journal of Medicine.1 Holz is a vitreoretinal surgeon and chair of the Department of Ophthalmology, University of Bonn, Germany.
This research may represent the first retinal prosthetic device treatment that may restore functional central vision to patients with GA due to AMD.1 The first new recently approved drug therapies for GA—pegcetacoplan injection (Syfovre; Apellis Pharmaceuticals) and avacincaptad pegol intravitreal solution (Izervay; Astellas Pharma Inc)—work by inhibiting the complement cascade and slowing disease progression.2-4 “However, no approved therapies, investigational approaches, or cell therapies have led to a meaningful improvement in vision,” Holz and colleagues noted.5-9
In the retinas of patients with GA, the photoreceptors, which are adversely affected by the disease, fail to convert the light entering the eye into electrical signals that would be transmitted to the brain, as in healthy eyes.10 The PRIMA implant is intended to replace the failed photoreceptors in affected eyes.11
The wireless device, which the investigators describe as a thin crystalline silicon array implanted subretinally within the atrophic lesion, measures 2 × 2 mm and is 30 µm thick (Figure). The array comprises 378 photovoltaic pixels 100 µm in width.12,13 In addition to the implant, the system includes a camera that is mounted on spectacles. The authors describe that the “PRIMA glasses capture images and project them, after processing, onto the implant with the use of near-infrared light (wavelength, 880 nm).” The function of the 378 pixels is to convert near-infrared light into electrical pulses to stimulate the retinal bipolar cells, which restores the flow of visual information,12 they explained.
The PRIMA system was tested preclinically,14-16 after which it was evaluated in a human clinical trial that included 5 patients with GA secondary to AMD to test its feasibility.17 The results showed that 3 of the 5 participants were able to recognize sequences of letters and had VA closely matching the maximal resolution of 20/420 allowed by the pixel width (100 μm).13 Four years after the implantation, the 3 patients read small fonts, with a mean Snellen VA of 20/135.17
The study under discussion—PRIMAvera (NCT04676854), an open-label, multicenter, prospective, single-group, baseline-controlled clinical trial—was conducted to evaluate the efficacy and safety of the device in patients with GA due to AMD.1 All patients, who had VA of 1.2 logMAR, were assessed at 6 and 12 months after implantation with and without the PRIMA glasses.1 The investigators related that their primary end points were meaningful VA improvement, which they defined as 0.2 logMAR or greater from baseline to month 12 after implantation, and the development of any serious adverse events that were associated with the procedure or device over the course of observation.1
Thirty-eight patients underwent implantation of the PRIMA device, and 32 completed the evaluation at 12 months.1 Among the other patients, 3 had died, 1 withdrew from the study, and 2 were not available for evaluation.1
Holz and colleagues reported on the 32 patients who were assessed at 12 months. “The PRIMA system led to a clinically meaningful improvement in VA from baseline in 26 [patients] (81%; 95% [CI], 64-93; P < .001),” they noted.1 They used multiple imputation to account for the missing data from the 6 patients, and based on that, they estimated that “80% (95% CI, 66-94; P < .001) of all participants would have had a clinically meaningful improvement at 12 months.”
Regarding adverse events, 26 serious events occurred in 19 patients; all were related to the implantation procedure solely or the procedure and the implant, but not to the implant alone. Twenty-one of these (81%) occurred within 2 months postoperatively, of which 20 (95%) resolved within 2 months after onset. Twenty-two events were mild or moderate in severity, while 4 were considered severe and included development of a full-thickness macular hole, ocular hypertension, retinal detachment, and proliferative vitreoretinopathy, they reported.
Based on the results, the researchers concluded, “The data at 12 months in this clinical study showed that the PRIMA subretinal implant restored meaningful central vision in persons with GA due to AMD, thus enabling the performance of visual tasks such as reading and writing. Although no retinal implants have been explanted in humans, the wireless design allows for replacement with higher-resolution next-generation implants18,19 or implantation of multiple arrays in a tiled pattern at the atrophic area with minimal incision.”20


Source: www.ophthalmologytimes.com
Author: | Date: 2025-12-29 11:00:00