Cataract
Share your love

VSY Biotechnology’s SVT Granted Patent in China
VSY Biotechnology registered a patent with the People’s Republic of China for the ‘Sinusoidal Vision Technology’ used in AcrivaUD Trinova Pro C Pupil Adaptive®, the world’s first and only sinusoidal trifocal intraocular lens. As a result of evaluations, office actions,…
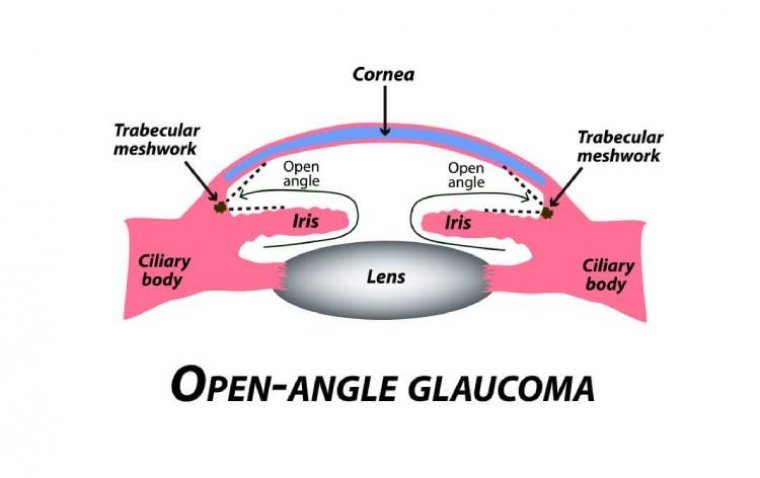
Primary Open Angle Glaucoma (POAG)
A subtype of glaucomas known as primary open angle glaucoma (POAG) is characterized by an open, seemingly normal anterior chamber angle and elevated intraocular pressure (IOP), with no additional underlying disease. Secondary glaucoma is a condition where the elevated IOP…

What is Photophobia and How to Manage it
Photophobia, or sensitivity to light, is a common condition characterized by discomfort or pain in response to exposure to light. While it can occur in individuals with healthy eyes, photophobia is often associated with underlying eye conditions or systemic diseases.…

Nordic Pharma Introduces Lacrifill Canalicular Gel for Dry Eye
Nordic Pharma announced the commercial launch of Lacrifill Canalicular Gel, aimed at addressing dry eye disease in the United States. This novel therapy, Lacrifill, is a cross-linked hyaluronic acid derivative that has received clearance from the FDA. Its primary function…
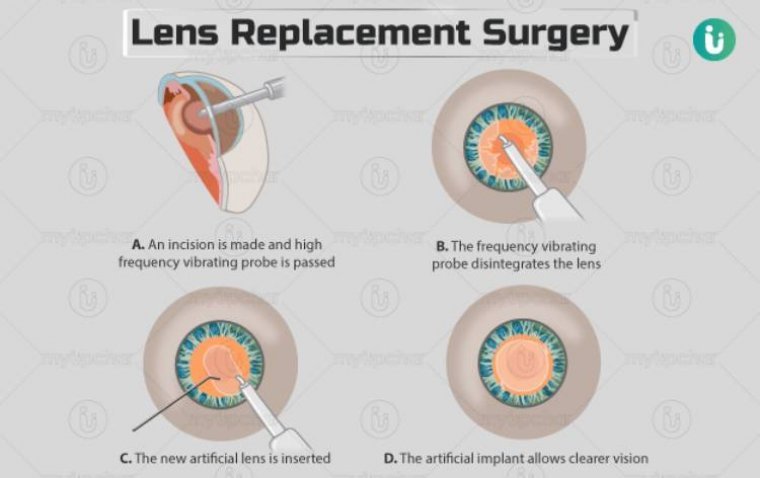
What Is Lens Replacement Surgery?
Lens replacement surgery, also known as Refractive Lens Exchange (RLE) or Clear Lens Extraction (CLE), is a procedure designed to improve vision by replacing the eye’s natural lens with an artificial one. This surgery is particularly beneficial for those who…
Your Complete Guide to Cataract Surgery
Cataract surgery is a safe, effective procedure that improves vision by removing cloudy lenses from the eyes and replacing them with artificial lenses. As technology advances and techniques improve, understanding the nuances of cataract surgery is crucial for anyone considering…

Melt Pharmaceuticals Launches MELT-300 Phase 3 Program
Melt Pharmaceuticals has completed the dosing of its first patient in its phase 3 program for MELT-300, marking a crucial step in evaluating the safety and efficacy of this pioneering product. MELT-300 is a non-intravenous, non-opioid tablet comprising fixed doses…

AAO’s Tool for Enhanced IOL Cataract Surgery
The U.S. Food and Drug Administration (FDA) has qualified the Assessment of IntraOcular Lens Implant Symptoms (AIOLIS), developed by the American Academy of Ophthalmology (AAO), as a Medical Device Development Tool (MDDT). This designation empowers medical device sponsors to employ…

Exclusive Interview with Prof. Dr. Oliver Findl
*If you are interested in becoming a part of our OBN Spotlight TV series, reach us at info@ophthalmologybreakingnews.com. Q1: As you reflect on the recent ESCRS 2023 Congress, could you share with us the key highlights and breakthroughs in cataract…

KDB GLIDE FDA 510(k) Clearance for Expanded Glaucoma Use
New World Medical announced a significant milestone with the receipt of 510(k) indication expansion from the U.S. Food and Drug Administration (FDA) for KDB (Kahook Dual Blade) GLIDE. This clearance allows for the reduction of intraocular pressure (IOP) in adult…





