Clinical Trial
Share your love

FDA approves carbachol–brimonidine eye drop for presbyopia following phase 3 BRIO trials | Ophthalmology Times
FDA approval of YUVEZZI marks the first dual-agent, daily eye drop option for adults seeking non-invasive presbyopia treatment. (Image credit: Ophthalmology Times) The FDA has approved a fixed-dose ophthalmic combination of carbachol and brimonidine tartrate (YUVEZZI; Tenpoint Therapeutics) for the…

FDA Type C meeting supports phase 2b/3 pathway for urcosimod in neuropathic corneal pain | Ophthalmology Times
(Image Credit: AdobeStock/OlegKachura) Editor’s Note: This content was generated with the assistance of AI. A US Food and Drug Administration (FDA) Type C meeting has clarified key elements of the late-phase development pathway for urcosimod, an investigational topical therapy for neuropathic…

Belite Bio completes enrollment in phase 2/3 DRAGON II trial for STGD1 | Ophthalmology Times
(Image Credit: AdobeStock/khunkornStudio) Belite Bio has completed enrollment in its phase 2/3 DRAGON II clinical trial evaluating tinlarebant for the treatment of Stargardt disease type 1 (STGD1). Tinlarebant is a novel oral therapy intended to reduce the accumulation of vitamin…
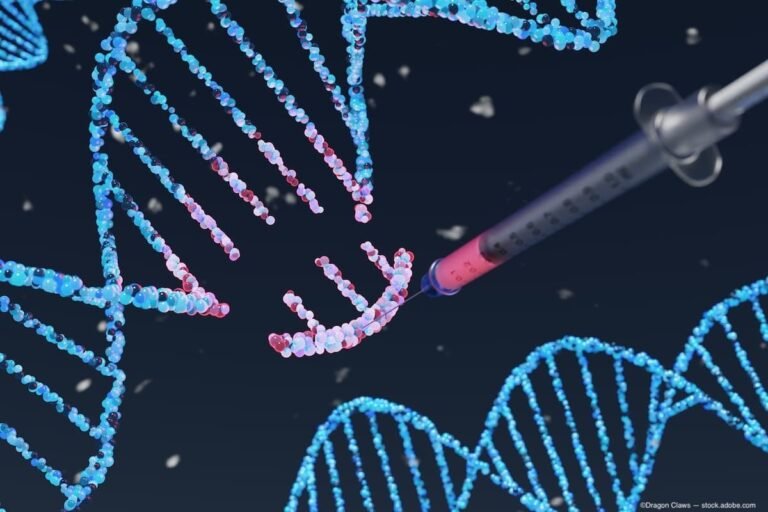
Opus Genetics launches phase 1/2 MERTK gene therapy trial | Ophthalmology Times
(Image Credit: AdobeStock/Dragon Claws) Opus Genetics has launched its clinical trial evaluating OPGx-MERTK gene therapy for MERTK-related retinitis pigmentosa (RP). The trial is funded through Abu Dhabi’s Healthcare Research and Innovation Fund. OPGx-MERTK is an investigational adeno-associated virus (AAV)-based gene…

Myra Vision treats first patient in ADAPT trial of Calibreye TGT Surgical System | Ophthalmology Times
(Image Credit: AdobeStock/andrew_shots) Myra Vision has treated the first patient in its ADAPT clinical study evaluating the Calibreye TGT Surgical System in patients with glaucoma. The ADAPT trial is a prospective, multicenter, nonrandomized, open-label study enrolling 70 refractory glaucoma patients…

Cloudbreak Pharma completes FDA end-of-phase-2 meeting for CBT-004 in pinguecula | Ophthalmology Times
(Image Credit: AdobeStock/pressmaster) Cloudbreak Pharma and the US Food and Drug Administration have completed an end-of-phase 2 meeting regarding the late-stage development of CBT-004 for the treatment of pinguecula. According to the company, this meeting helped to provide a “clear…

FDA clears Novaliq’s Investigational New Drug application for NOV05 | Ophthalmology Times
(Image Credit: AdobeStock/Stock Contributor) The US Food and Drug Administration (FDA) has cleared Novaliq’s Investigational New Drug (IND) application for NOV05, enabling the initiation of the EYETAC phase II clinical trial (NCT07285070) in patients with non-infectious anterior uveitis (NIAU). NOV05,…

Nanoscope’s gene therapy, MCO-010 receives Sakigake and Orphan Drug Designations in Japan | Ophthalmology Times
(Image Credit: AdobeStock/sezerozger) Nanoscope Therapeutics has received Pioneering Regenerative Medical Product (Sakigake) and Orphan Drug designations for MCO-010 from Japan’s Ministry of Health, Labour and Welfare (MHLW), making it the first retinal gene therapy to do so in Japan. Sakigake designation is Japan’s…

SpyGlass Pharma randomizes first patients in 2 phase 3 clinical trials of the BIM-IOL System | Ophthalmology Times
(Image Credit: AdobeStock/Pixel-Shot) SpyGlass Pharma has randomized the first patients in its 2 registrational phase 3 clinical trials of the Bimatoprost Drug Pad-IOL System (BIM-IOL System) for the lowering of intraocular pressure (IOP) in patients with open-angle glaucoma (OAG) or…

Q&A: Evolving approaches to geographic atrophy management | Ophthalmology Times
(Image Credit: AdobeStock/Prostock-studio) Management of geographic atrophy (GA) has changed with the introduction of retinal therapies aimed at slowing disease progression and with greater use of imaging for diagnosis and monitoring. These developments have raised practical questions about patient selection,…





