
Newsletter Subscribe
Enter your email address below and subscribe to our newsletter


Enter your email address below and subscribe to our newsletter

(Image Credit: AdobeStock/Who is Danny) AAVantgarde and AGC Biologics have entered into an agreement in which AGC Biologics will provide Good Manufacturing Practice manufacturing for AAVantgarde’s dual-vector gene therapies for inherited retinal disorders, AAVB-039 and AAVB-081.1 Most recently, AAVantgarde Bio…

(Image Credit: AdobeStock/Julien Tromeur) Editor’s Note: This content was generated with the assistance of AI. At AAO 2025, the conversation around retinal disease treatment moved decisively toward the future—one defined by senolytic agents, next-generation gene and optogenetic therapies, disease-modifying approaches…

(Image Credit: AdobeStock/ronstik) Ocular Therapeutix has randomized the first patient in its HELIOS-3 phase 3 registrational program for AXPAXLI (or OTX-TKI) for the treatment of non-proliferative diabetic retinopathy (NPDR). The company describes AXPAXLI as an investigational, bioresorbable, intravitreal hydrogel incorporating…
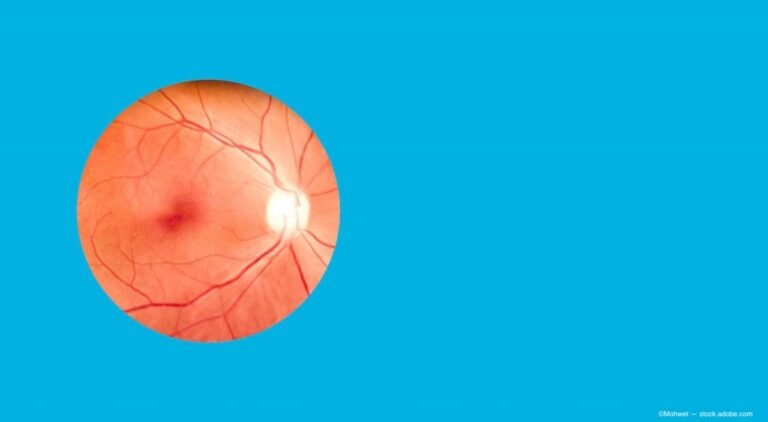
(Image Credit: AdobeStock/Mohwet) Editor’s Note: This content was generated with the assistance of AI. AAO 2025 showcased some of the most significant leaps in retinal imaging and disease surveillance the field has seen in over a decade. Three presentations—delivered by…

The US Food and Drug Administration (FDA) has approved aflibercept injection 8 mg (EYLEA HD) from Regeneron Pharmaceuticals for the treatment of patients with macular edema following retinal vein occlusion (RVO) with up to every 8-week dosing after an initial monthly…

(Image credit: ©peopleimages.com/AdobeStock) The Data Safety Monitoring Committee (DSMC) recently completed its second scheduled review of EyePoint Pharmaceuticals’ ongoing phase 3 trial evaluating DURAVYU for the treatment of wet AMD (wAMD), EyePoint announced.1 The DSMC concluded that both the LUGANO…
(Image Credit: AdobeStock/Anna) Precise Bio has successfully treated a patient with PB-001, the company’s 3D-bio-printed corneal implant, at Rambam Medical Center in Haifa, Israel. The procedure was conducted as part of the company’s ongoing phase 1 clinical trial. PB-001 is…

The Viewpoints series “Reimagining Dry Eye Management: Targeting Tear Function for Sustained Relief” convenes leading experts in ophthalmology and optometry to discuss evolving perspectives on dry eye disease (DED). Under the moderation of Kendall Donaldson, MD, MS, the panel—comprising Cecelia…

The Viewpoints series “Reimagining Dry Eye Management: Targeting Tear Function for Sustained Relief” convenes leading experts in ophthalmology and optometry to discuss evolving perspectives on dry eye disease (DED). Under the moderation of Kendall Donaldson, MD, MS, the panel—comprising Cecelia…
A new study from researchers at EPFL introduces a promising non-invasive brain stimulation technique aimed at restoring visual function in stroke patients with hemianopia. The treatment combines visual training with targeted neuromodulation, offering a novel approach to visual rehabilitation in…