
Newsletter Subscribe
Enter your email address below and subscribe to our newsletter


Enter your email address below and subscribe to our newsletter

Alcon has announced the U.S. launch of PRECISION7, a groundbreaking contact lens designed for a convenient one-week replacement cycle. Incorporating the advanced 7-day ACTIV-FLO system, these lenses offer up to 16 hours of comfort and clear vision, even on the…

Osheru announced the successful completion of a $4.7 million funding round aimed at accelerating the development and commercialization of its Ziplyft device for blepharoplasty surgery. The funding round was oversubscribed and led by a consortium of investors including individuals, family…

Mumbai-based ENTOD Pharmaceuticals has unveiled a novel advancement in ophthalmic care with the introduction of EyeBS, a new eye drop preservative system. EyeBS is specifically designed to supplant traditional detergent-based preservatives such as Benzalkonium Chloride (BAK) and Polyquaternium-1, offering a…

PulseMedica has successfully raised a total of $12 million in their recent pre-Series A financing round, marking a significant milestone in their pursuit of innovative eye floater diagnostic and treatment technologies. Accelerating Clinical Validation and Expansion Efforts This funding round…
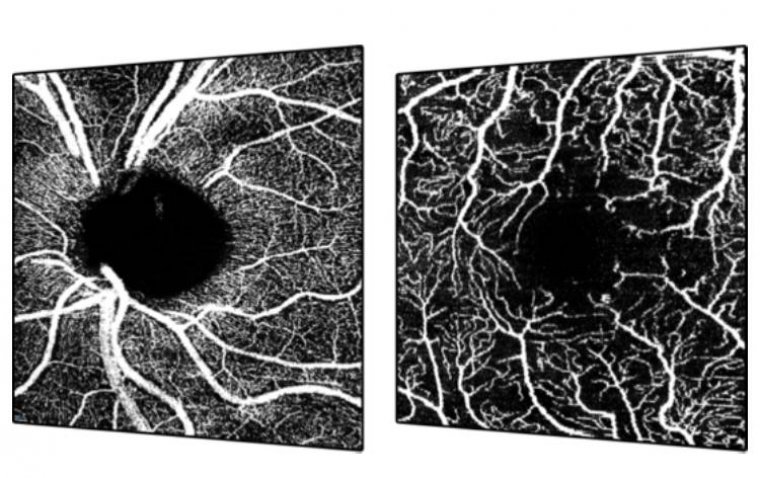
Heidelberg Engineering announced the FDA clearance of its upgraded SPECTRALIS® OCTA Module featuring SHIFT technology. This advancement promises to significantly reduce acquisition time by 50%, marking a pivotal development in clinical efficiency and diagnostic precision within ophthalmology. Enhancing Clinical Utility…

Sydnexis, Inc., a pioneering biopharmaceutical company dedicated to addressing pediatric myopia progression, has appointed Perry J. Sternberg as its Chief Executive Officer and as a member of its Board of Directors. Sternberg, a seasoned pharmaceutical and biotech executive with over…

VSY Biotechnology GmbH, a leading company with innovative ophthalmic solutions, is now accepting submissions for Ophthalmology Star Awards (OSA) 2024 from eye care professionals worldwide. The awards aim to celebrate and promote novel research and advancements in ophthalmology, with winners…

NVISION Eye Centers, a prominent provider of eye care services nationwide, announced a strategic collaboration with Parkhurst NuVision of San Antonio, Texas, through its subsidiary NVision Clinical Research, LLC. This partnership aims to advance clinical trials in ophthalmology drug development,…

4D Molecular Therapeutics (4DMT) announced the FDA clearance of its investigational new drug application (IND) for 4D-175, an R100 vector-based intravitreal genetic medicine intended for treating geographic atrophy (GA) in patients. Phase 1 GAZE Clinical Trial Details The phase 1…

Ikerian has received European Union Medical Device Regulation (EU-MDR) certificate for its RetinAI Discovery digital data platform and AI-based models. These products have been classified as Class IIa medical devices, affirming their adherence to stringent regulatory standards for healthcare technology.…