
Newsletter Subscribe
Enter your email address below and subscribe to our newsletter


Enter your email address below and subscribe to our newsletter

In a significant move for the ophthalmology sector, Carl Zeiss Meditec announced the successful acquisition of 100% shares of the Dutch Ophthalmic Research Center (DORC), a transaction that has now received all necessary regulatory approvals. This acquisition from Eurazeo SE,…

Melt Pharmaceuticals has successfully completed a significant financial milestone, securing approximately $24 million through its Series B Preferred Stock financing round. This latest funding round saw contributions from both new and returning investors, setting the stage for the accelerated development…

CSI Dry Eye Software, a leading innovator in the field of artificial intelligence for eye care, has officially announced the release of its latest software update, Version 5.0. This update marks a significant leap forward in the company’s mission to…

Viatris Inc. announced the commercial launch of RYZUMVI™ (phentolamine ophthalmic solution) 0.75% in the United States. This treatment is the only FDA-approved eye drop available in the U.S. market designed to reverse pharmacologically-induced mydriasis, commonly caused by adrenergic agonists like…

The Glaucoma Research Foundation (GRF) has recently released a 48-minute audiobook version of their comprehensive 40-page educational guide, “Understanding and Living with Glaucoma.” This project aims to extend the reach of their essential information to those affected by glaucoma, especially…

Aurion Biotech has dosed the first Canadian participant in its phase 1/2 clinical trial, known as ABA-1, CLARA. The trial investigates the efficacy and safety of AURN001, a novel cell therapy aimed at treating corneal edema resulting from corneal endothelial…

ABVC BioPharma, Inc. has entered into a significant global licensing agreement with ForSeeCon Eye Corporation (FEYE), marking a pivotal moment for the clinical-stage biopharmaceutical company known for its developments in ophthalmology, CNS, and oncology The agreement focuses on ABVC’s ophthalmology…
In a significant development for patients with geographic atrophy (GA) secondary to age-related macular degeneration (AMD), Iveric Bio, a subsidiary of Astellas Pharma, has been granted a unique, permanent J-code by the Centers for Medicare & Medicaid Services (CMS) for…
In a pivotal development in the field of ophthalmology, Nanodropper Inc announced that the results from a clinical trial evaluating the Nanodropper® Adaptor have been published in the esteemed journal, Ophthalmology. The study’s findings indicate that there was no clinically…
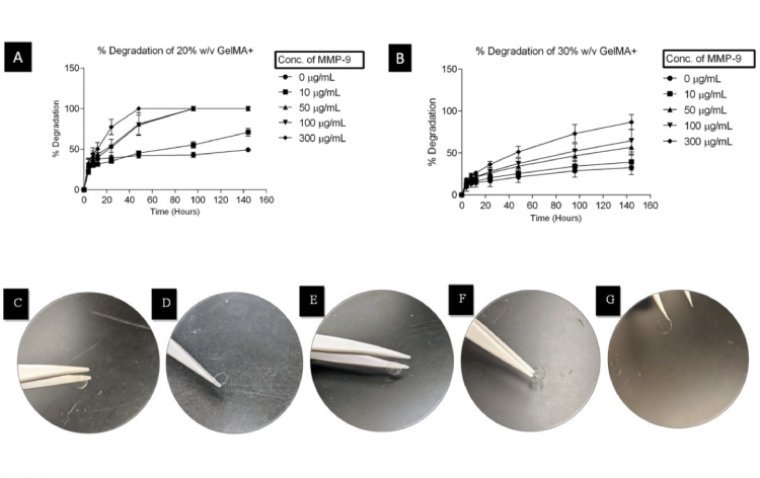
Scientists at the University of Waterloo have made a groundbreaking development in eye care technology by creating a new type of contact lens that not only serves as a protective bandage for corneal wounds but also delivers healing drugs in…