Patient Education
Share your love

Dr. Arshad Khanani Joins Ophtea as Chief Medical Advisor
Opthea Limited announced Dr. Arshad M. Khanani, MD, MA, FASRS, as its new Chief Medical Advisor. Dr. Khanani, renowned in retina specialization and clinical research, brings a wealth of expertise from his tenure at Sierra Eye Associates and his academic…

Evmann Holding Eyes Expansion in Ophthalmology at ESCRS
Evmann Investments Holding BV is taking a proactive approach to strengthening its investment footprint in the ophthalmology sector. The Netherlands-based holding company, with operations spanning over 93 countries via its German subsidiary VSY Biotechnology GmbH, is setting a new course…

Scientist Awarded $300K for AMD Research
Dr. Rajendra Apte, MD, PhD, has been honored with the Research to Prevent Blindness (RPB)/American Macular Degeneration Foundation (AMDF) Catalyst Award for Innovative Research Approaches for Age-Related Macular Degeneration (AMD). This award, amounting to $300,000, underscores Dr. Apte’s dedication and…

MACUSTAR Extends Systematic AMD Research
The MACUSTAR consortium, led by the Department of Ophthalmology at the University Hospital Bonn (UKB), is extending its Europe-wide clinical study on age-related macular degeneration (AMD). The primary focus of the extension is to garner further insights into the intermediate…

Lupin Launches Bromfenac Ophthalmic Solution in the US
Lupin Limited announced the launch of Bromfenac Ophthalmic Solution, 0.075%. The product has received the green light from the United States Food and Drug Administration (U.S. FDA), marking a crucial milestone for the company’s expansion in the American pharmaceutical market.…

Opthea Completes Enrollment for Wet AMD Trial
Opthea has reached a significant milestone in its quest to combat wet age-related macular degeneration (AMD) with the completion of patient enrollment in the COAST phase 3 pivotal clinical trial. The trial investigates sozinibercept (OPT-302), a promising vascular endothelial growth…

VSY Biotechnology’s 100% Glistening-Free IOL Material
VSY Biotechnology GmbH is pushing the boundaries of ophthalmology with its cutting-edge 100% Glistening-Free IOL Material. In a statement, Andreas Kühnel, the Managing Director at VSY Biotechnology GmbH, expressed his excitement about the groundbreaking innovation, saying, “This innovative solution, developed…
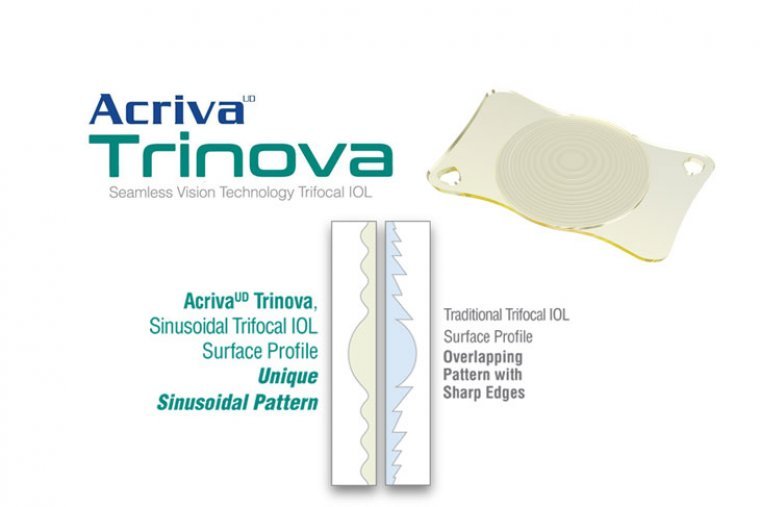
A Novel Approach To Trifocal IOLs
AcrivaUD Trinova is the world’s first and only sinusoidal trifocal IOL manufactured with Sinusoidal Vision Technology (SVT). Its sinusoidal optic design eliminates the sharp steps (diffractive rings) found with traditional trifocal IOLs. The smoothly varying surface profile derived from the…

ESCRS 2023 Recap: Biotech Companies That Stand Out
As OBN, we had the privilege of participating in the ESCRS Congress 2023 held in Vienna, where we witnessed the remarkable efforts of various companies dedicated to enhancing patient outcomes. In this exclusive article, we present a compilation of ten…
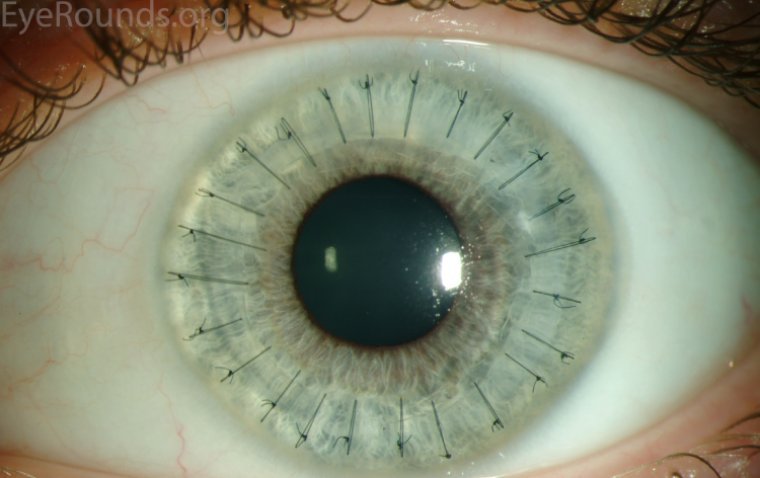
What is Penetrating Keratoplasty and How to Perform it
What is Penetrating Keratoplasty? Penetrating Keratoplasty (PKP), also known as a full-thickness corneal transplant, is a significant ophthalmic surgical procedure aimed at restoring visual acuity in patients with corneal diseases. This procedure involves the replacement of the entire corneal tissue…





