
Newsletter Subscribe
Enter your email address below and subscribe to our newsletter


Enter your email address below and subscribe to our newsletter

CorneaGen, a leading provider of ocular tissue, announced the commercial launch of Corneal Tissue Addition for Keratoplasty (CTAK), a novel solution for corneal contouring in keratoconic eyes. Developed in collaboration with CTAK LLC and Ziemer, CTAK offers patient-ready, gamma-irradiated sterile…

BIOPHTA, a preclinical biotech company, has successfully closed a Seed round, raising €6.5 million to advance its pioneering technology into clinical development for the treatment of glaucoma and macular edema. This significant funding injection, led by prominent European venture capital…

Azura Ophthalmics has reached a significant milestone with the enrollment of the first patient in the ASTRO study—a phase 3 clinical trial evaluating the efficacy and safety of AZR-MD-001 in patients grappling with clinical signs and symptoms of meibomian gland…

Sightpath Medical, a leading provider of mobile and fixed ophthalmic surgical services, has recently completed the acquisition of PennVista, a renowned LASIK services business. PennVista specializes in offering cutting-edge equipment and skilled technicians to surgery centers and hospitals across Pennsylvania,…
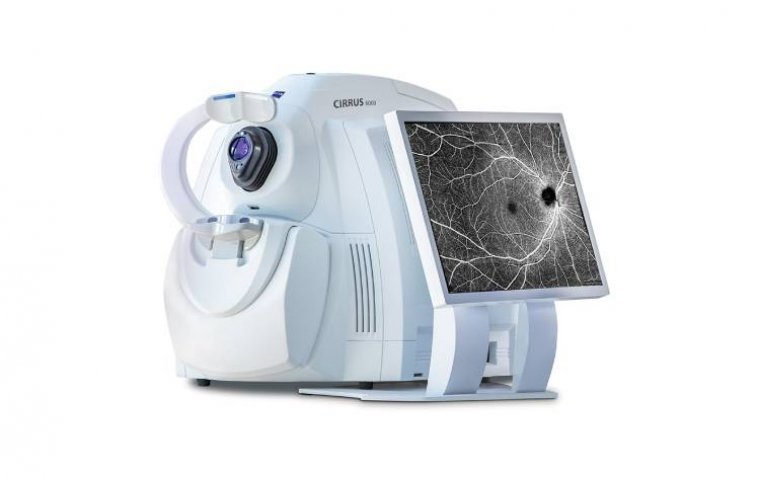
Zeiss Medical Technology has introduced a series of enhancements to the Cirrus 6000, promising a more streamlined and data-centric workflow for ophthalmologists. This update boasts the largest OCT reference database in the US market and fortified cybersecurity features. With recent…

The U.S. Food and Drug Administration (FDA) has qualified the Assessment of IntraOcular Lens Implant Symptoms (AIOLIS), developed by the American Academy of Ophthalmology (AAO), as a Medical Device Development Tool (MDDT). This designation empowers medical device sponsors to employ…

The increasing prevalence of digital devices in children’s lives has sparked a growing concern among parents, educators, and healthcare professionals about the potential impacts on eye health. As screens become an integral part of education, entertainment, and social interaction, understanding…

In a significant development for the medical community, Drug Farm announced that the U.S. Food and Drug Administration (FDA) has granted clearance for the Investigational New Drug (IND) application of DF-003. This novel drug, hailed as a first-in-class oral medication,…
The USC Roski Eye Institute is making significant strides in the realm of Thyroid Eye Disease (TED) treatment through pioneering clinical trials exploring new medications. TED, a condition characterized by bulging eyes and potential vision impairment, has traditionally necessitated multiple…

Exonate announced the expansion of its Board with the appointment of Dr. Rafiq Hasan as Non-Executive Director. This strategic move aims to bolster the company’s expertise in ophthalmology, particularly within the diabetic eye disease sector, as it advances its lead…