
Newsletter Subscribe
Enter your email address below and subscribe to our newsletter


Enter your email address below and subscribe to our newsletter

The California Institute for Regenerative Medicine (CIRM) has awarded a $6 million, two-year grant to researchers at the USC Dr. Allen and Charlotte Ginsburg Institute for Biomedical Therapeutics and the USC Roski Eye Institute. This funding will advance preclinical studies…

Adverum Biotechnologies has released promising 52-week topline results from its Phase 2 LUNA trial and new 4-year data from the Phase 1/2 OPTIC study for its gene therapy candidate Ixo-vec (AAV.7m8-aflibercept), targeting wet age-related macular degeneration (AMD). Transformative Potential of…

Emmecell has announced positive results from its U.S. multicenter Phase 1 trial evaluating EO2002, an innovative non-surgical cell therapy for corneal edema. The randomized, double-masked trial demonstrated significant improvements in best corrected visual acuity (BCVA) and reductions in central corneal…

Ocugen has released encouraging preliminary findings from the Phase 1 portion of its Phase 1/2 OCU410 ArMaDa clinical trial, focused on treating geographic atrophy (GA) secondary to dry age-related macular degeneration (AMD). The results demonstrate the potential of OCU410 as…

Key Takeaways • Gildeuretinol (ALK-001) has received Rare Pediatric Disease and Fast Track designations, alongside prior Breakthrough Therapy and Orphan Drug statuses, emphasizing its potential to transform treatment for Stargardt disease. • Data from the TEASE-1…

Laser-assisted in situ keratomileusis (LASIK) has become one of the most effective and safe methods for correcting refractive error with laser corneal intervention, as evidenced by the long and large number of publications in the peer-reviewed literature. In recent years,…

Samsung Bioepis and Biogen have announced that the European Commission (EC) has approved Opuviz 40 mg/mL for injection. This biosimilar, also known as SB15, references the widely used treatment Eylea (aflibercept). The approval encompasses a range of retinal conditions, providing…

Aldeyra Therapeutics has announced a major milestone as the FDA formally accepted its resubmitted New Drug Application (NDA) for reproxalap, a topical ocular therapy for the treatment of dry eye disease. Alongside this regulatory progress, Aldeyra has expanded its exclusive…

In late October, Medicare introduced proposed Local Coverage Determinations (LCDs) that aimed to impose restrictions on Minimally Invasive Glaucoma Surgery (MIGS) procedures. These LCDs were met with significant concern and opposition from medical professionals, patient advocacy groups, and the ophthalmology…
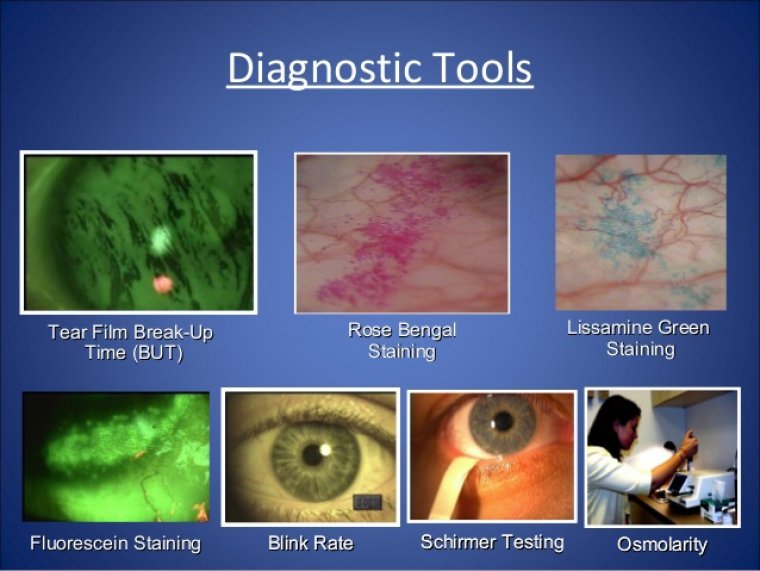
Dry eye disease (DED), also known as keratoconjunctivitis sicca, is one of the most common ophthalmic conditions, affecting hundreds of millions of people worldwide. Recent technological advances and research targeting DED have led to the emergence of new definitions and…