Case Reports
Share your love

AAO 2025: Diana Do, MD, reviews the real-world use of aflibercept 8 mg | Ophthalmology Times
Note: Video captions are generated with the assistance of AI and may contain errors. Diana Do, MD, a professor of ophthalmology and vice chair of Clinical Affairs at Stanford University’s Byers Eye Institute, presented research on aflibercept 8 mg in…

Aurion Biotech releases positive 12-month results from phase 1/2 CLARA trial | Ophthalmology Times
(Image Credit: AdobeStock/tonstock) Aurion Biotech has released positive 12-month results from its phase 1/2 CLARA trial evaluating the safety, efficacy, and tolerability of AURN001 in patients with corneal edema secondary to corneal endothelial dysfunction. In the CLARA trial, a double-masked,…

FDA grants breakthrough therapy designation to Nacuity’s NPI-001 for retinitis pigmentosa | Ophthalmology Times
(Image Credit: AdobeStock) The US Food and Drug Administration (FDA) has granted Nacuity Pharmaceuticals’ N-acetylcysteine amide tablets (NPI-001) breakthrough therapy designation. NPI-001 is Nacuity’s investigational therapy for the treatment of patients with retinitis pigmentosa (RP). It is a GMP-grade formulation…

AAVantgarde’s AAVB-039 gene therapy to treat Stargardt disease receives orphan drug status from FDA | Ophthalmology Times
(Image Credit: AdobeStock/artacet) The US Food and Drug Administration (FDA) has granted AAvantgarde’s AAVB-039 Orphan Drug Designation (ODD) and has also received Clinical Trial Authorisation (CTA) approval from the UK’s Medicines and Healthcare Products Regulatory Agency (MHRA). AAVB-039 is an…

Charles Miller, MD, PhD, on DME research and the initiation of the phase 2b/3 BRUNELLO trial | Ophthalmology Times
Image credit: AdobeStock/MrAshi On September 4, 2025, Merck and EyeBio announced that the companies have initiated the Phase 2b/3 BRUNELLO trial (NCT06571045) to evaluate the candidate MK-3000, formerly known as EYE103 for the treatment of patients with diabetic macular edema…
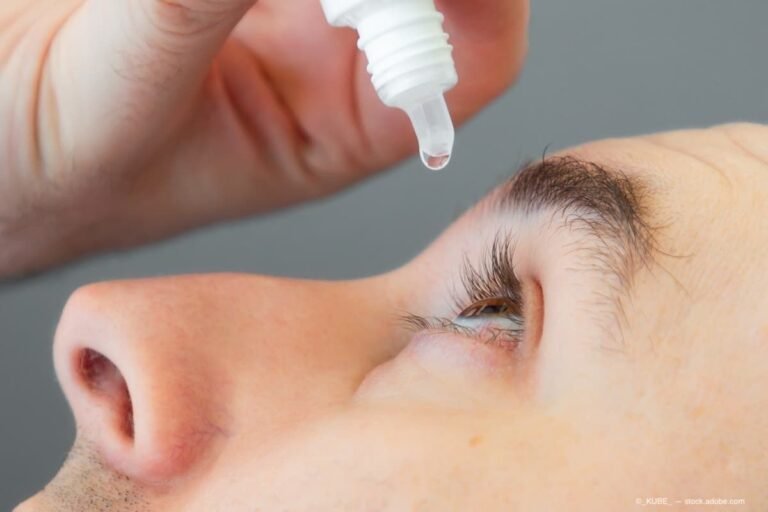
Vizz officially launches in the United States | Ophthalmology Times
(Image Credit: AdobeStock) Following a US Food and Drug Administration approval back in July 2025, LENZ Therapeutics has announced that VIZZ (aceclidine ophthalmic solution) 1.44% is now commercially available.1 The first and only FDA-approved aceclidine-based eye drop is for the…

David Lally discusses the ARCHER trial and the optimistic outcomes fueling future research | Ophthalmology Times
Note: Video captions are generated with the assistance of AI and may contain errors. David Lally, MD, presented at the 2025 Retina Society meeting, discussing the ARCHER study (NCT04656561) of ANX007 for the treatment of age-related macular degeneration (AMD) and…

Q&A: Mario Romano discusses temperature during vitrectomy | Ophthalmology Times
Photo of Mario Romano at EURETINA 2025 Mario Romano, MD, PhD, a researcher from Humanitas University in Italy, discusses work on temperature control during vitrectomy. His research, supported by the Italian Ministry of Research, reveals significant temperature drops during eye…

Q&A: Nadia Waheed on the role of OCT in the future of ophthalmology | Ophthalmology Times
Photo of Nadia Waheed at the 2025 EURETINA meeting held in Paris, France Ultra High Resolution OCT is revolutionizing retinal imaging, offering unprecedented insights into eye diseases. Nadia Waheed, MD, MPH, from Tufts University School of Medicine discusses how this…

Q&A: Patricio Schlottmann discusses results of AVONELLE-X faricimab extension trial | Ophthalmology Times
Photo of Patricio Schlottmann at the 2025 EURETINA meeting in Paris, France Patricio Schlottmann, MD, a retina specialist from Buenos Aires, Argentina, discusses the AVONELLE-X extension study of faricimab, a treatment for wet age-related macular degeneration (AMD). The study, spanning…





