
Newsletter Subscribe
Enter your email address below and subscribe to our newsletter


Enter your email address below and subscribe to our newsletter

Emmetrope Ophthalmics, a leader in cell-based therapies for severe eye diseases, today announced the completion of the final dose administration in its U.S. clinical trial for EO2002, targeting the treatment of corneal edema. EO2002 Advances Toward Phase 3 This milestone…

Inflammasome Therapeutics has officially dosed the first patient in a pioneering clinical trial aimed at treating geographic atrophy (GA) resulting from age-related macular degeneration (AMD). This trial, a first-in-class study for a sustained release implant, is sponsored by the University…
Regeneron has received FDA approval for Eylea HD (aflibercept 8 mg) in a significant development anticipated to substantially extend dosing intervals for patients with retinal diseases such as wet age-related macular degeneration (AMD), diabetic macular edema (DME), and diabetic retinopathy (DR).…

LumiThera has been granted a small business innovative research (SBIR) phase 2 grant from the National Institute of Health (NIH), totaling up to $2.3 million in funding over a span of 2 years. This NIH/NEI grant is geared towards supporting…
Repeat corticosteroid injections were found to be more effective in improving vision for individuals with persistent or recurrent uveitis-related macular edema compared to methotrexate or ranibizumab intravitreal injections, according to a recent clinical trial. The corticosteroid treatment exhibited superior results…
Parent company Astellas Pharma announced that Iveric Bio has received FDA approval for Izervay (avacincaptad pegol intravitreal solution) to treat geographic atrophy (GA) secondary to age-related macular degeneration (AMD). As a new complement C5 inhibitor, Izervay stands out as the sole…

Naegis Pharmaceuticals Inc., a life sciences company specializing in inflammation and ophthalmology, announced the successful closure of a financing round to advance its lead compound, N0651, towards a Phase 2 clinical trial. This funding will support the compound’s progression through…
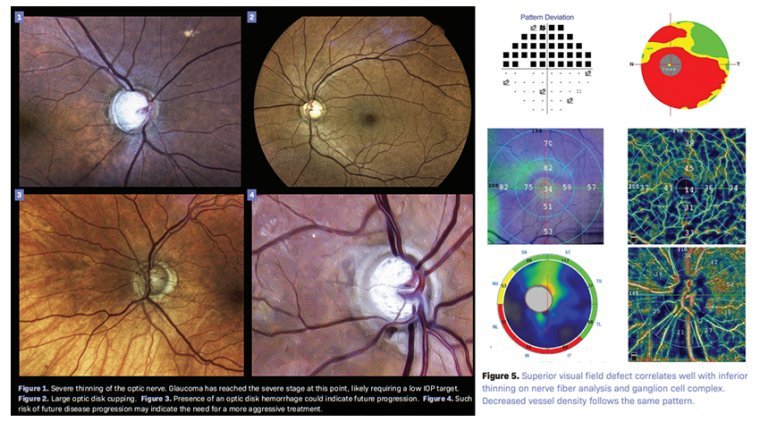
Glaucomatous optic neuropathy is characterized by progressive loss of retinal ganglion cells (RGCs) and their axons and leads to measureable structural and functional damage to the optic nerve, visual impairment, and blindness. The primary site of injury is thought to be…

Ocugen has officially completed dosing in the second cohort of its phase 1/2 ArMaDa trial of OCU410, a novel gene therapy designed to treat geographic atrophy (GA). The therapy, known as adeno-associated viral vector 5 human RORA (AAV5-hRORA), is being…
A novel multi-ingredient supplement significantly improves ocular symptom severity and tear production in patients with dry eye disease: results from a randomized, placebo-controlled clinical trial Frontiers Source: Author: | Date: 2024-04-23 07:00:00 Source: Author: | Date: 2024-04-23 07:00:00