
Newsletter Subscribe
Enter your email address below and subscribe to our newsletter


Enter your email address below and subscribe to our newsletter

Ocular Therapeutix announced positive topline results from its phase 1 HELIOS study, which evaluated the safety, tolerability, and efficacy of Axpaxli versus a sham control in patients with moderately severe to severe nonproliferative diabetic retinopathy (NPDR) without diabetic macular edema…

Kiora Pharmaceuticals announced that it has secured a grant from the Choroideremia Research Foundation (CRF) to support novel clinical trial endpoints for inherited retinal diseases (IRDs). The grant will fund the validation of the Multiluminence Orientation & Mobility (MLOM™) suite…

Aldeyra Therapeutics announced its clinical development strategy aimed at the resubmission of a New Drug Application (NDA) for its topical ocular 0.25% reproxalap. This investigational RASP modulator is poised for the treatment of dry eye disease, pending approval from the…

Aurion Biotech has dosed the first Canadian participant in its phase 1/2 clinical trial, known as ABA-1, CLARA. The trial investigates the efficacy and safety of AURN001, a novel cell therapy aimed at treating corneal edema resulting from corneal endothelial…

Opus Genetics recently completed the first stage of its innovative clinical trial, focusing on a new treatment for patients suffering from Leber congenital amaurosis (LCA) due to biallelic mutations in the LCA5 gene. The trial, which is in the phase…

BlueRock Therapeutics LP, a clinical-stage cell therapy company and a subsidiary of Bayer, in partnership with Foundation Fighting Blindness, has announced their collaboration to introduce a new cohort to the Foundation’s ‘Uni-Rare natural history study’. This initiative focuses on individuals…
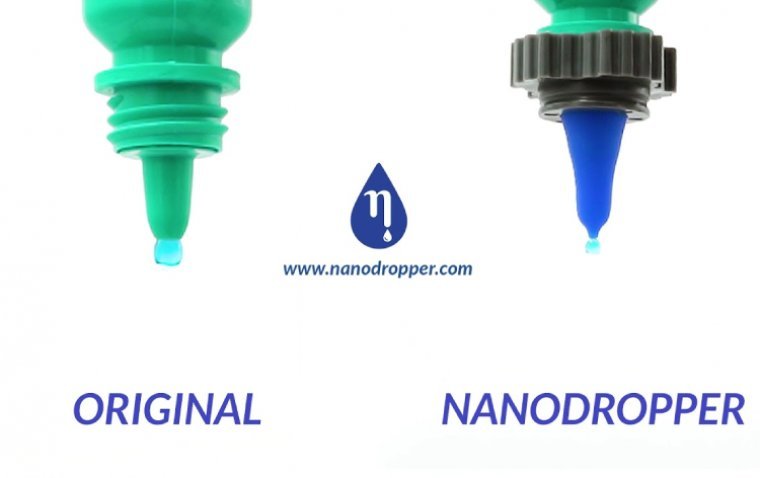
In a pivotal development in the field of ophthalmology, Nanodropper Inc announced that the results from a clinical trial evaluating the Nanodropper® Adaptor have been published in the esteemed journal, Ophthalmology. The study’s findings indicate that there was no clinically…

In a recent phase II clinical trial conducted by the National Eye Institute (NEI), part of the National Institutes of Health, the antibiotic minocycline, known for its anti-inflammatory properties, was tested for its efficacy in slowing down the progression of…

Innovent Biologics announced that its second Phase 2 clinical trial of efdamrofusp alfa (R&D code: IBI302), a high-dose recombinant human VEGFR-Fc-Human CR1 fusion protein injection, has achieved its primary endpoint in treating Chinese subjects with neovascular age-related macular degeneration (nAMD).…
In a groundbreaking era of medical research, scientists and clinicians are spearheading novel gene therapies that hold promise for transforming the landscape of eye disease treatment. These cutting-edge interventions address a spectrum of conditions ranging from Usher Syndrome and retinoschisis…