
Newsletter Subscribe
Enter your email address below and subscribe to our newsletter


Enter your email address below and subscribe to our newsletter

Skye Bioscience’s phase 2a clinical trial of its SBI-100 ophthalmic emulsion in patients with primary open-angle glaucoma (POAG) or ocular hypertension (OHT) has failed to meet its primary endpoint for lowering intraocular pressure (IOP). Consequently, the company announced its decision…

Melt Pharmaceuticals has completed the dosing of its first patient in its phase 3 program for MELT-300, marking a crucial step in evaluating the safety and efficacy of this pioneering product. MELT-300 is a non-intravenous, non-opioid tablet comprising fixed doses…

Harrow, a leading pharmaceutical company, has revealed the encouraging results from its ESSENCE‑2 open-label extension (OLE) clinical study for Vevye (cyclosporine ophthalmic solution) 0.1%, a novel treatment for dry eye disease (DED). Study Design and Outcomes: ESSENCE-2 OLE Analysis The…

Azura Ophthalmics has reached a significant milestone with the enrollment of the first patient in the ASTRO study—a phase 3 clinical trial evaluating the efficacy and safety of AZR-MD-001 in patients grappling with clinical signs and symptoms of meibomian gland…
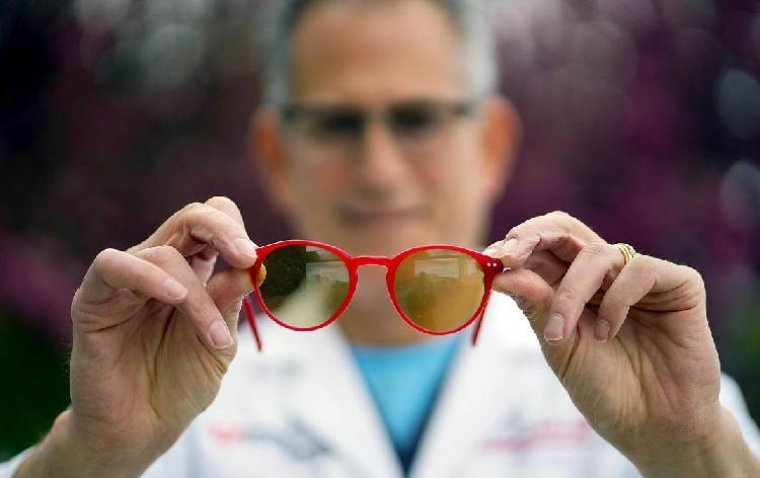
This spring, individuals grappling with migraines find relieve with a novel solution for managing light sensitivity, thanks to new eyeglass lens technology developed by a physician-scientist at the University of Utah John A. Moran Eye Center. The Impact of Migraine…

In a significant development for the medical community, Drug Farm announced that the U.S. Food and Drug Administration (FDA) has granted clearance for the Investigational New Drug (IND) application of DF-003. This novel drug, hailed as a first-in-class oral medication,…

Exonate announced the expansion of its Board with the appointment of Dr. Rafiq Hasan as Non-Executive Director. This strategic move aims to bolster the company’s expertise in ophthalmology, particularly within the diabetic eye disease sector, as it advances its lead…

A study led by a University at Buffalo researcher has the potential to revolutionize the treatment of retinopathy of prematurity (ROP), a condition that can lead to blindness in low birthweight premature infants if left untreated. Published in eClinicalMedicine, the…
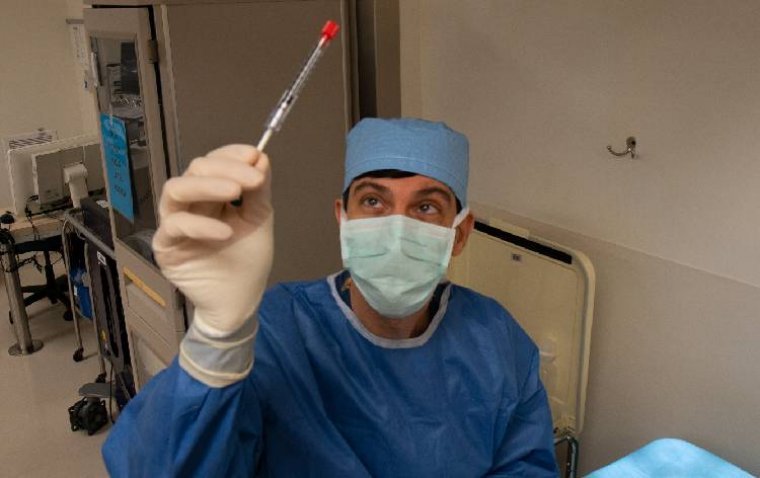
In a landmark clinical trial, CRISPR-Cas9 gene editing has proven safe and effective in treating a form of inherited blindness, marking a significant milestone in genetic medicine. The multi-site BRILLIANCE trial, involving the Perelman School of Medicine at the University…

EyePoint Pharmaceuticals announced the topline results from its phase 2 PAVIA clinical trial, which evaluated the efficacy of Duravyu (vorolanib intravitreal insert), formerly known as EYP-1901, in treating non-proliferative diabetic retinopathy (NPDR). The trial revealed that Duravyu did not achieve…