
Newsletter Subscribe
Enter your email address below and subscribe to our newsletter


Enter your email address below and subscribe to our newsletter

Ocugen has released encouraging preliminary findings from the Phase 1 portion of its Phase 1/2 OCU410 ArMaDa clinical trial, focused on treating geographic atrophy (GA) secondary to dry age-related macular degeneration (AMD). The results demonstrate the potential of OCU410 as…

Sun Pharmaceutical has shared new Phase 4 clinical trial data, showcasing the efficacy of Cequa (cyclosporine ophthalmic solution) 0.09% in treating dry eye disease (DED). The results reveal that Cequa provides sustained improvements in patients whose symptoms were inadequately managed…

MediPrint Ophthalmics has revealed positive findings from its Phase 2b clinical trial for LL-BMT1, a 3D-printed, drug-eluting contact lens designed to treat mild to moderate glaucoma. These results, presented by Ian Ben Gaddie, OD, FAAO, at the American Academy of…

Okyo Pharma announced its plan to initiate a phase 2 clinical trial for OK-101 in the treatment of neuropathic corneal pain (NCP), scheduled to begin in the third quarter of 2024. This one-year study follows promising results from preclinical animal…

NexThera has officially submitted a Phase 1/2a Investigational New Drug (IND) application to the U.S. Food and Drug Administration (FDA) for NT-101, an innovative non-invasive eye drop treatment targeting wet age-related macular degeneration (wet AMD). Proprietary Delivery Platform: Advancing Treatment…

PulseMedica has successfully raised a total of $12 million in their recent pre-Series A financing round, marking a significant milestone in their pursuit of innovative eye floater diagnostic and treatment technologies. Accelerating Clinical Validation and Expansion Efforts This funding round…
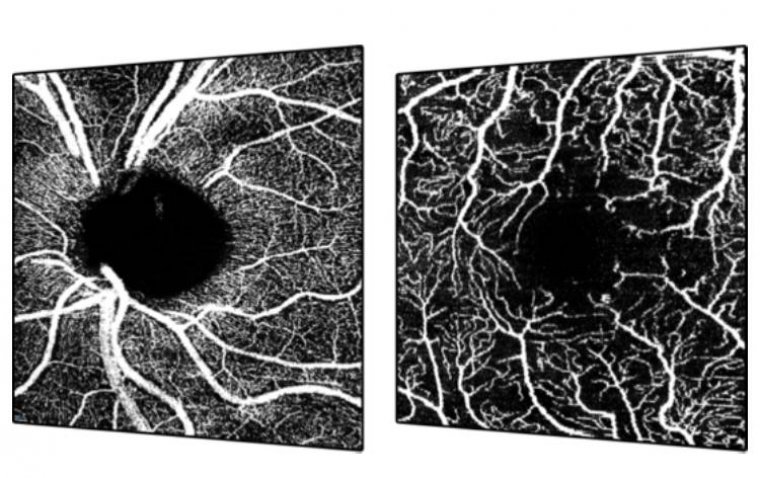
Heidelberg Engineering announced the FDA clearance of its upgraded SPECTRALIS® OCTA Module featuring SHIFT technology. This advancement promises to significantly reduce acquisition time by 50%, marking a pivotal development in clinical efficiency and diagnostic precision within ophthalmology. Enhancing Clinical Utility…

Outlook Therapeutics announced that the UK Medicines and Healthcare products Regulatory Agency (MHRA) has granted marketing authorization for Lytenava (bevacizumab gamma) for the treatment of wet age-related macular degeneration (AMD) in the UK. Lytenava is the first and only authorized…
Beacon Therapeutics has successfully raised $170 million in Series B funding, marking a significant step forward in advancing its portfolio of ophthalmic gene therapies. The funds are earmarked to support the ongoing clinical development of Beacon’s lead candidate, AGTC-501, designed…

A new artificial intelligence (AI) system developed by researchers from UCL and Moorfields Eye Hospital could potentially revolutionize the recruitment process for clinical trials aimed at treating Geographic Atrophy (GA), an advanced form of dry age-related macular degeneration (AMD) that…