
Newsletter Subscribe
Enter your email address below and subscribe to our newsletter


Enter your email address below and subscribe to our newsletter
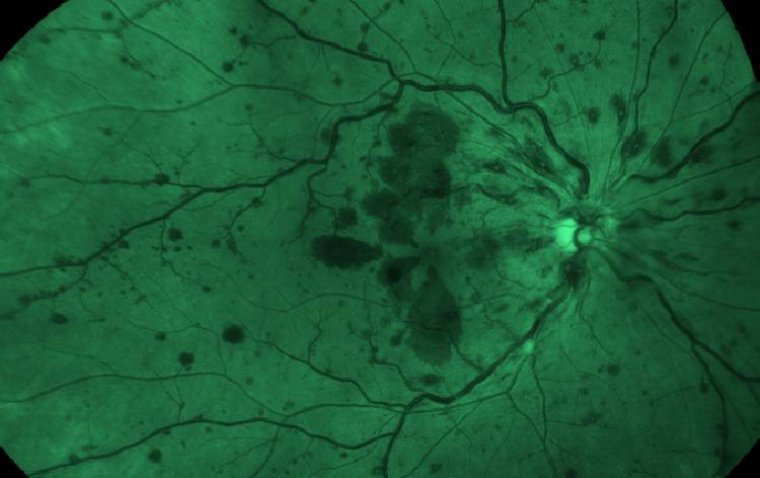
Dr. Fu-Shin Yu, a professor of Ophthalmology, Visual and Anatomical Sciences at Wayne State University School of Medicine, has been awarded a significant grant from the National Eye Institute of the National Institutes of Health. The five-year grant, totaling $2,167,882,…

Glenmark Pharmaceuticals has received final approval by the FDA for Brimonidine Tartrate and Timolol Maleate Ophthalmic Solution, 0.2%|0.5% for glaucoma treatment. This solution has been recognized by the FDA as bioequivalent and therapeutically equivalent to Combigan® Ophthalmic Solution, 0.2%|0.5% by…

Notal Vision announced that the FDA has granted De Novo authorization for its patient self-operated Scanly Home OCT device. Designated by the FDA as a breakthrough device for patients suffering from wet age-related macular degeneration (AMD), Scanly Home OCT aims…
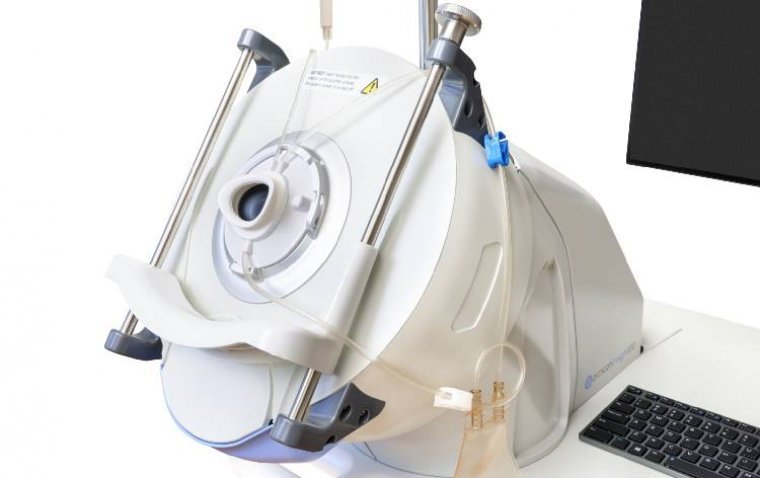
ArcScan, a global medical technology company dedicated to revolutionizing ophthalmic care, marked a significant milestone with the announcement of the National Medical Products Administration (NMPA) approval of its flagship product, the ArcScan Insight® 100, for use in China. Addressing the…

Humonix Biosciences, a pioneering biotechnology company, has recently named Dr. Karen Torrejon as its new Chief Executive Officer. Dr. Torrejon, who originally founded the company in 2014, will now lead its operations as it transitions into a broader, science-driven platform.…

EyeCRO recently announced the results from the inaugural clinical application of its MiDROPS (Microemulsion Drug Ocular Penetration System) technology, presented by Telios Pharma at the Association for Research in Vision and Ophthalmology (ARVO) meeting held in Seattle, WA. The presentation,…

Oculis has completed enrollment in its multicenter, randomized, double-blind, placebo-controlled phase 2 ACUITY trial evaluating the safety and tolerability of OCS-05 in patients with Acute Optic Neuritis (AON). Topline results are expected in Q4 2024. The phase 2 ACUITY study…

Researchers have developed a new type of contact lens that contains a community of lubricant-producing bacteria to combat contact lens-associated dry eye, a common issue that leads many to discontinue use. The findings were published in Advanced Materials. Innovative Solution…

Eluminex Biosciences announced the acceptance of its EB-105 Investigational New Drug (IND) application by the FDA. This significant milestone marks a pivotal moment in the advancement of medical science, particularly in the realm of Diabetic Macular Edema (DME) treatment. EB-105:…

Topcon Healthcare has announced an investment in RetiSpec, marking the beginning of a collaboration aimed at commercializing RetiSpec’s innovative technology. This partnership intends to bridge the gap between neurology and eye care, potentially transforming how Alzheimer’s disease is detected and…