Industry News
Share your love

Tenpoint Therapeutics Launches with $70M Financing
Tenpoint Therapeutics, a biotechnology company focused on vision-restoring engineered cell-based therapeutics and in vivo reprogramming for degenerative ocular diseases, has announced its launch with a $70 million Series A financing. The funding will be used to develop the company’s regenerative…

Naegis Pharma Funds Novel Eye Drug
Naegis Pharmaceuticals Inc., a life sciences company specializing in inflammation and ophthalmology, announced the successful closure of a financing round to advance its lead compound, N0651, towards a Phase 2 clinical trial. This funding will support the compound’s progression through…

2023 Recap: Top 10 Contact Lenses and Technologies
In the ever-evolving landscape of ophthalmology, 2023 has proven to be a year of groundbreaking advancements in contact lens technology. From lenses that adapt to UV and temperature changes to intelligent lenses capable of diagnosing and treating glaucoma, the top…
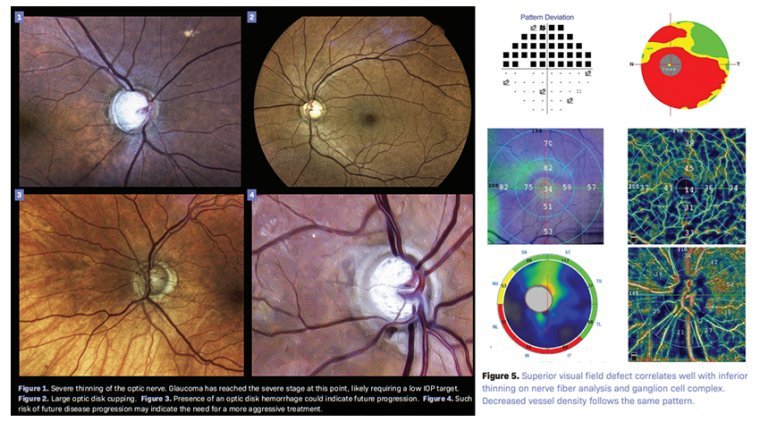
Glaucomatous Optic Neuropathy & Latest Treatments
Glaucomatous optic neuropathy is characterized by progressive loss of retinal ganglion cells (RGCs) and their axons and leads to measureable structural and functional damage to the optic nerve, visual impairment, and blindness. The primary site of injury is thought to be…

SpyGlass Pharma Secures $90M in Funding
SpyGlass Pharma has successfully closed a Series C financing round, raising $90 million. RA Capital Management led the funding round, joined by existing investors New Enterprise Associates (NEA) and Vensana Capital, as well as new investors Samsara BioCapital and Vertex…
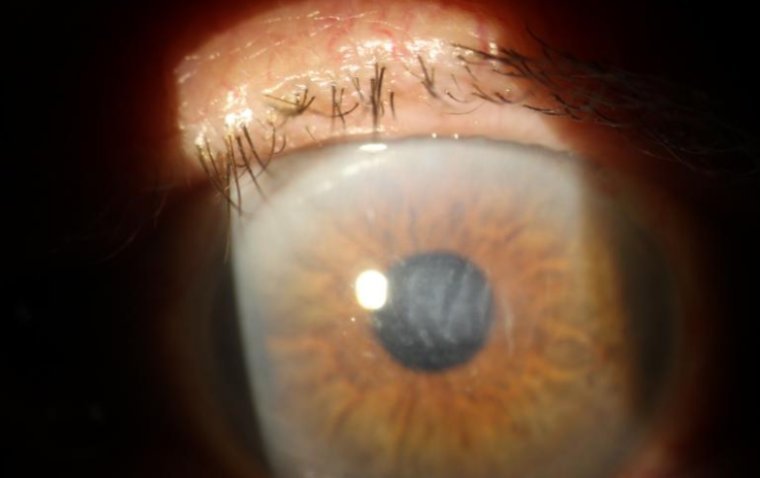
What Is Corneal Edema and How to Manage It
What Is Corneal Edema? Corneal edema refers to the swelling of the cornea, the transparent, dome-shaped layer at the front of the eye. This swelling typically results from an abnormal accumulation of fluid within the corneal layers, disrupting its usual…

OMNI Surgical System Greatly Reduces Medication Usage
Sight Sciences released study findings demonstrating that patients with glaucoma who had undergone procedures using their OMNI Surgical System technology had a greater average reduction in the usage of intraocular pressure-lowering medication compared to those who had received only minimally…
Belkin’s Eagle Device FDA Cleared for Laser Trabeculoplasty
Belkin Vision has received FDA 510(k) clearance for its Eagle device, a Q-switched, 532 nm-wavelength, frequency-doubled Nd:YAG laser developed for selective laser trabeculoplasty (SLT). The Eagle is positioned by Belkin as the first and only contactless laser for glaucoma, offering…

FDA Warms 8 Companies for Unapproved Eye Items
On Monday, the US Food and Drug Administration (FDA) took action by sending warning letters to several companies, including CVS and Walgreens, due to concerns regarding the manufacturing and marketing of unapproved eye products. According to the FDA, these products…

Carl Zeiss Meditec to Acquire D.O.R.C.
Carl Zeiss Meditec AG announced that they have entered into an agreement to acquire 100% of the shares in D.O.R.C. Dutch Ophthalmic Research Center (International) B.V. from Eurazeo SE, based in Paris, France. The deal is worth €985 million (nearly…





