
Newsletter Subscribe
Enter your email address below and subscribe to our newsletter


Enter your email address below and subscribe to our newsletter

RevOpsis Therapeutics has announced the successful closure of its first seed funding round, securing a total of $16.5 million. This funding is set to advance the development and commercialization of treatments for chronic multifactorial diseases through the company’s innovative, fully…

LEP Biomedical announced pre-seed funding of €100,000 ($107,000) over two 6-month periods, which the company says will allow for further preclinical milestones in the development of the HyaGuard platform. The funding was made through Enterprise Ireland. Innovative Design of HyaGuard…

Ocugen has officially completed dosing in the second cohort of its phase 1/2 ArMaDa trial of OCU410, a novel gene therapy designed to treat geographic atrophy (GA). The therapy, known as adeno-associated viral vector 5 human RORA (AAV5-hRORA), is being…
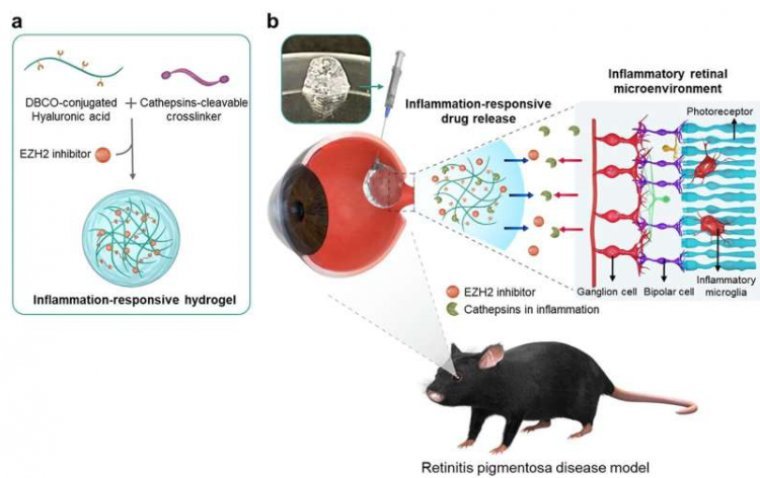
A breakthrough study has unveiled a promising advancement in the treatment of incurable eye diseases such as age-related macular degeneration and retinitis pigmentosa. Researchers have successfully integrated anti-inflammatory drugs into a hydrogel, aiming to combat inflammation in the retina while…

Ocular Therapeutix announced positive topline results from its phase 1 HELIOS study, which evaluated the safety, tolerability, and efficacy of Axpaxli versus a sham control in patients with moderately severe to severe nonproliferative diabetic retinopathy (NPDR) without diabetic macular edema…

Caplin Steriles Limited (Caplin), a subsidiary of Caplin Point Laboratories Limited, has received final approval from the United States Food and Drug Administration (USFDA) for its Abbreviated New Drug Application (ANDA) for Ketorolac Tromethamine Ophthalmic Solution 0.5%. This product is…

Medevise Consulting, a strategic consultancy specializing in the ophthalmic sector, announced that it has received ISO13485 certification. This certification marks a significant achievement on the company’s third anniversary, underscoring its commitment to quality and operational excellence alongside its adeptness in…

Kiora Pharmaceuticals announced that it has secured a grant from the Choroideremia Research Foundation (CRF) to support novel clinical trial endpoints for inherited retinal diseases (IRDs). The grant will fund the validation of the Multiluminence Orientation & Mobility (MLOM™) suite…

The U.S. Department of Justice (DOJ) has initiated a lawsuit against Regeneron Pharmaceuticals under the False Claims Act (FCA), accusing the biotechnology company of fraudulently inflating Medicare reimbursement rates. According to the DOJ, Regeneron submitted false average sales price reports…

Lenz Therapeutics announced positive topline results from its Phase 3 CLARITY study, evaluating two investigational formulations, LNZ100 and LNZ101, aimed at treating presbyopia. The study’s findings showcase the effectiveness and safety of these formulations, particularly highlighting LNZ100 (1.75% aceclidine) for…