
Newsletter Subscribe
Enter your email address below and subscribe to our newsletter


Enter your email address below and subscribe to our newsletter

Aviceda Therapeutics announced that the FDA has granted clearance for the IND for AVD-104. This clearance will allow the company to proceed with the initiation of Phase 2 clinical trials of its leading intravitreal ocular asset for the treatment of…
In this article, we explore five significant milestones in the history of ophthalmology that have shaped the way we approach eye health and treatment. The ancient Egyptians, known for their advanced civilization and remarkable contributions to various fields, including medicine,…

Aldeyra Therapeutics announced the receipt of a Complete Response Letter from the U.S. Food and Drug Administration (FDA) in response to the New Drug Application (NDA) for reproxalap, an investigational drug candidate designed for the treatment of dry eye disease.…
In children with amblyopia, often referred to as “lazy eye,” one eye exhibits weaker vision than the other due to various factors. This weaker eye may struggle with focusing, misalignment due to strabismus, or vision obstruction from conditions like cataracts…

A recent study published in Nature Medicine has unveiled promising insights into a potential breakthrough in the treatment of Diabetic Macular Edema (DME). The study suggests that an experimental drug, part of a new class of therapeutics called “senolytics,” could…
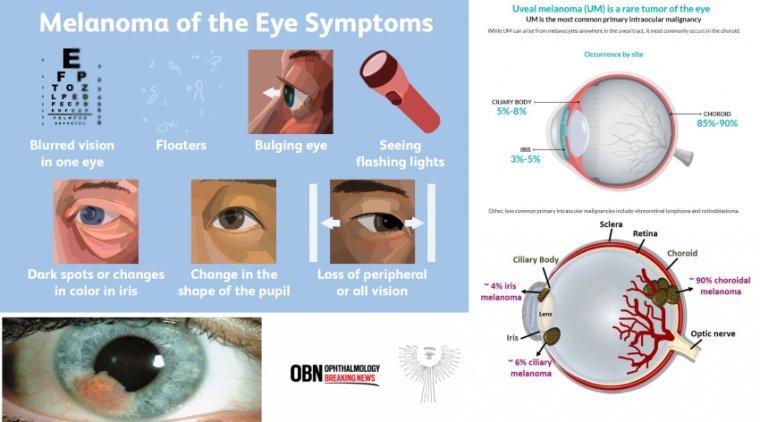
Melanoma of the uveal tract (iris, ciliary body, and choroid), though rare, is the most common primary intraocular malignancy in adults. The mean age-adjusted incidence of uveal melanoma in the United States is approximately 4.3 new cases per million people,…

4D Molecular Therapeutics (4DMT) announced positive interim data from the Phase 2 PRISM clinical trial, marking a significant stride in the treatment of wet age-related macular degeneration (wet AMD). Arshad M. Khanani, MD, MA, FASRS, presented the data at the…

Mireca Medicines has been awarded a $989,000 ‘Translational Research Acceleration Program Award’ from the Foundation Fighting Blindness, enabling the company to advance preclinical development of its lead product, MM238. Recognizing Mireca’s pivotal role in targeting the cGMP pathway for treating…

Formosa Pharmaceuticals announced a strategic licensing agreement with Cristália Produtos Químicos Farmacêuticos to secure exclusive commercialization rights for APP13007 (clobetasol propionate ophthalmic nanosuspension, 0.05%) in Brazil. APP13007 is designed for the treatment of inflammation and pain following ocular surgery and…

In a strategic move to advance myopia management as the standard of care, the World Council of Optometry (WCO) has teamed up with CooperVision to host its inaugural Latin America-based virtual event, “Myopia Management: From Theory to Practice.” Scheduled for…