
Newsletter Subscribe
Enter your email address below and subscribe to our newsletter


Enter your email address below and subscribe to our newsletter

The FDA has released a significant update for the pharmaceutical industry, unveiling a revised draft guidance titled “Quality Considerations for Topical Ophthalmic Drug Products.” This comprehensive guidance addresses crucial quality considerations for various ophthalmic drug products, including gels, ointments, creams,…
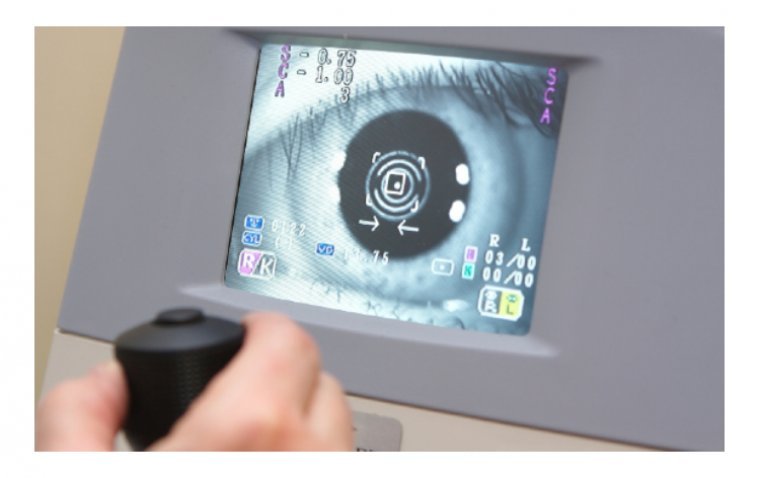
Medical technology has advanced significantly in recent years, providing numerous benefits to patients and healthcare professionals. In the field of ophthalmology, innovative medical devices have been developed that have revolutionized the way eye conditions are diagnosed and treated. Here are…

Azura Ophthalmics reported positive top-line efficacy and safety outcomes from a phase 2 investigation of AZR-MD-001 in individuals experiencing Contact Lens Discomfort (CLD) due to challenges in comfortably wearing lenses and demonstrating signs of meibomian gland dysfunction (MGD). The trial…
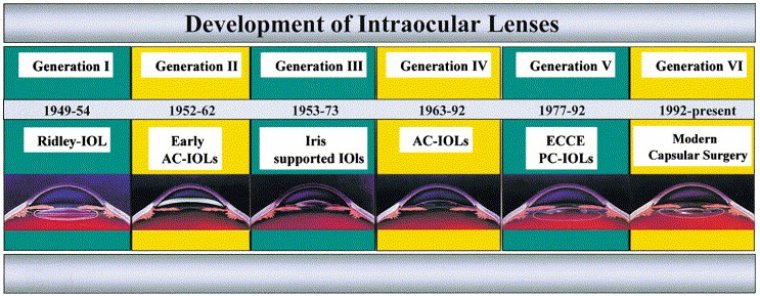
Cataract is the most prevalent ophthalmic disease. Treatment of cataracts has been practiced for centuries using various surgical and nonsurgical procedures. However, avoidance of complications and attainment of high-quality postoperative visual rehabilitation were difficult in the years before the introduction…
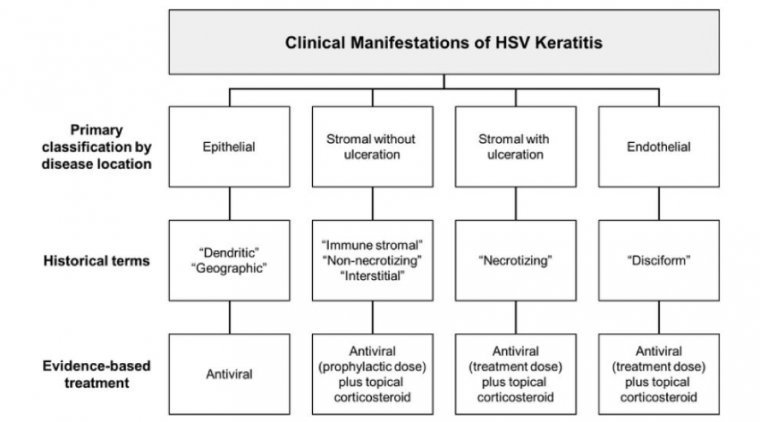
Trifluridine eye drop, acyclovir (ACV) ointment, ganciclovir gel, and oral ACV are still the main therapeutic agents. Cryopreserved amniotic membrane has been recently used as an adjuvant treatment. Resistance to ACV has become a concerning issue. The animal models of…
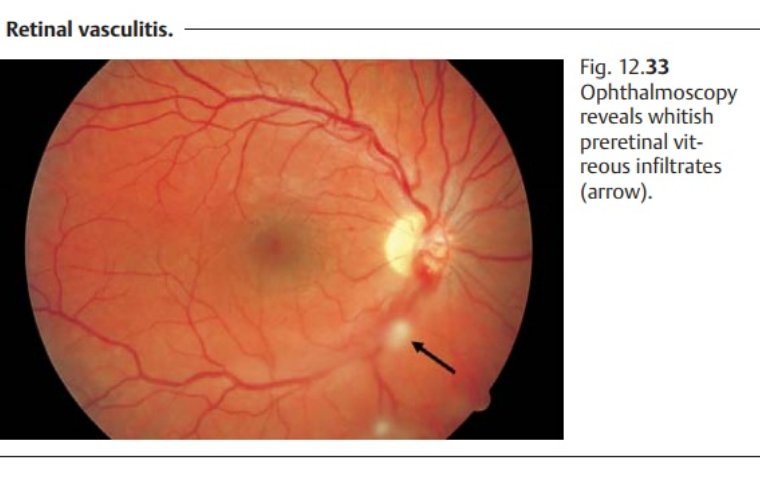
Retinal vasculitis is an inflammatory condition affecting the blood vessels of the retina. This inflammation can disrupt the normal blood flow, leading to complications that may compromise visual function. Pathophysiology In retinal vasculitis, inflammatory cells such as neutrophils, macrophages, and…

VSY Biotechnology, a leading company in ophthalmic innovations, made a notable presence at the 10th Evolving Practice of Ophthalmology Middle East Conference (EPOMEC 2023) in Dubai. The conference, held from 23-25 November, marked a significant moment for VSY Biotechnology as…

The US Food and Drug Administration’s (FDA) Center for Drug Evaluation and Research (CDER) has issued a warning letter to e-commerce giant Amazon.com regarding the introduction of unapproved ophthalmic drug products into interstate commerce. The FDA’s warning, delivered in a…

Bayer announced on Friday that the European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) has recommended the approval of a high-dose formulation of the renowned eye drug Eylea for the treatment of wet age-related macular degeneration and…

VSY Biotechnology GmbH is proud to announce the groundbreaking development of Varliciment, a revolutionary anti-VEGF agent for the treatment of age-related macular degeneration (AMD). Varliciment distinguishes itself from existing anti-VEGF agents like brolucizumab, namely Novartis’ Beovu, by being non-generic. This…