
Newsletter Subscribe
Enter your email address below and subscribe to our newsletter


Enter your email address below and subscribe to our newsletter

Bausch + Lomb has officially launched Miebo (perfluorohexyloctane ophthalmic solution) for the treatment of dry eye disease (DED) in the United States. Earlier in May, Bausch + Lomb and Novaliq received FDA approval for Miebo, marking it as the first…

Development of active glasses for mitigating presbyopia has become an active area of research, facilitated by the miniaturization of focus-tunable lenses as well as the increased processing power of modern computers. Alcon the global leader in eye care dedicated to…

The global ophthalmic sutures market size is expected to reach USD 532.8 million by 2026, expanding at a CAGR of 6.4% over the forecast period, according to a new report by Grand View research, Inc. Key factors driving the market…

Cataract surgery is the most frequently performed operative procedure worldwide, typically concludes with the implantation of an artificial intraocular lens (IOL) to correct aphakia (absence of the crystalline lens). The surgery—which involves removing the eye’s clouded lens and replacing it…

Lupin Limited, a global pharmaceutical company, has officially received approval from the US FDA for its Abbreviated New Drug Application for Bromfenac Ophthalmic Solution, 0.09%. This approval allows Lupin to introduce a generic counterpart to Bausch & Lomb Inc.’s Bromday…

The Medicines and Healthcare products Regulatory Agency (MHRA) has recently issued a National Patient Safety Alert due to reports of increased intraocular pressure in patients recently implanted with EyeCee One preloaded and EyeCee One Crystal preloaded intraocular lenses (IOLs), which…

Caeregen Therapeutics announced that it has received a $1.4 million Phase 2 Small Business Innovation Research (SBIR) grant from the National Institutes of Health (NIH) and National Eye Institute (NEI). The grant has been awarded to further the advancement of…

After the initial approval of Apellis Pharmaceuticals’ geographic atrophy drug Syfovre (pegcetacoplan injection), six cases of retinal vasculitis have been reported. The ASRS Research and Safety in Therapeutics (ReST) Committee issued a notification disclosing these safety concerns to ASRS members,…
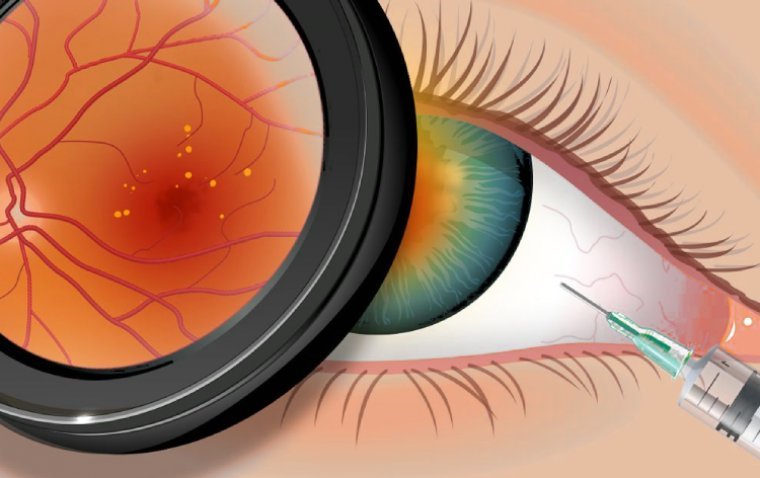
A recent study conducted by Columbia University researchers suggests that newly developed eye drops could potentially offer a more effective and comfortable treatment option for a common eye disease currently managed through eye injections. The eye disease in question is…

The European Commission has approved a revision to the EU Medical Device Regulation (MDR) to streamline the Unique Device Identifier (UDI) requirements for specific products, starting with contact lenses. Once implemented, the amendment will enable contact lens manufacturers to assign…