
Newsletter Subscribe
Enter your email address below and subscribe to our newsletter


Enter your email address below and subscribe to our newsletter
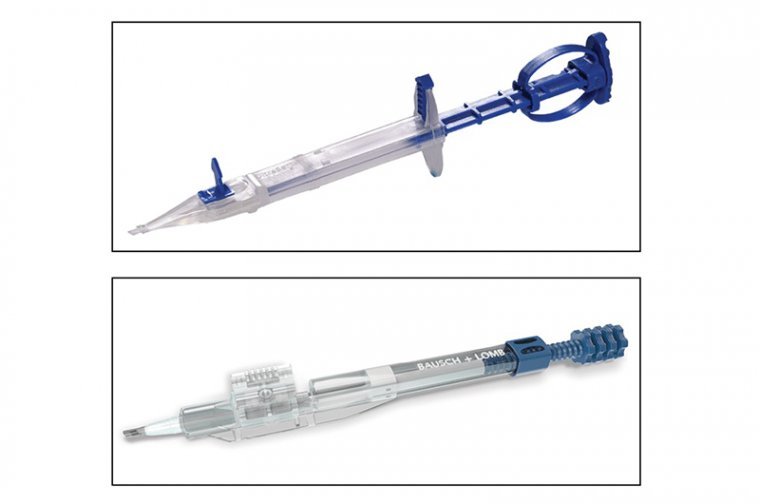
Preloaded injectors provide a number of benefits, including making the procedure safer by reducing the possibility of endophthalmitis from contamination and avoiding potential mistakes such as loading the lens incorrectly. It is certainly more efficient for the nursing or technical…
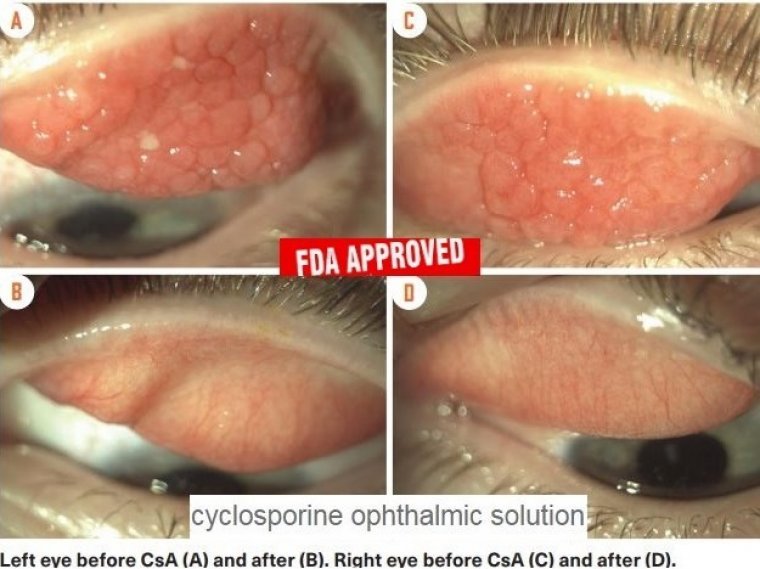
Verkazia 0.1%) eye drops containing cyclosporine have been authorized by the US Food and Drug Administration (FDA) for the treatment of vernal keratoconjunctivitis (VKC) in both adult and juvenile populations. The surface of the eyes in patients with the uncommon,…

TelomEYE Pharmaceuticals Corporation, working on a new generation of ophthalmologic therapies, announced today the completion of a successful $1 million post-seed funding round, valuing the company at $10 million USD. The round was co-led by healthcare mogul Mehmet Ali Aydınlar,…

Tarsus Pharmaceuticals has appointed Jeff Farrow as both Chief Financial Officer and Chief Strategy Officer, effective immediately. Farrow replaces Leo Greenstein, who had been CFO since 2020 but is leaving the company to pursue other professional interests. Farrow has over…

Bausch + Lomb Corporation has recently launched the StableVisc™ cohesive ophthalmic viscosurgical device (OVD) and the TotalVisc™ Viscoelastic System in the United States. These new products offer eye surgeons dual-action protection during cataract surgery, expanding the range of options available.…

Nicox SA announced that its exclusive Chinese partner, Ocumension Therapeutics, has submitted a new drug application (NDA) seeking approval for commercialization of Zerviate (cetirizine ophthalmic solution), 0.24%, in China. The intended use of Zerviate is for the treatment of ocular…

Horizon Therapeutics has announced positive and statistically significant topline results from its randomized, double-masked, placebo-controlled Phase 4 clinical trial evaluating TEPEZZA for the treatment of adults with chronic thyroid eye disease (TED) and low CAS, a measure of disease activity.…

The FDA has approved Apellis Pharmaceuticals’ Syfovre (pegcetacoplan) for the treatment of geographic atrophy (GA) secondary to age-related macular degeneration (AMD). This marks the first time that patients with GA have an FDA-approved treatment option, which is a significant milestone…

Johnson & Johnson Vision announced a series of new products and innovations at the 2019 American Academy of Ophthalmology (AAO) Annual Meeting in San Francisco, CA, USA. The company announced the US Food & Drug Administration (FDA) approval of the…

Leinfelden – Echterdingen, Germany March 19, 2021, VSY Biotechnology GmbH, an innovation-driven company with its progressive R&D activities in ophthalmology, visco supplementation in orthopedy and medical esthetics, announced today an agreement with WAKAMOTO PHARMACEUTICAL CO., LTD. (hereinafter refer to…