Pharma & Drugs
Share your love

OMNI Surgical System Greatly Reduces Medication Usage
Sight Sciences released study findings demonstrating that patients with glaucoma who had undergone procedures using their OMNI Surgical System technology had a greater average reduction in the usage of intraocular pressure-lowering medication compared to those who had received only minimally…
Belkin’s Eagle Device FDA Cleared for Laser Trabeculoplasty
Belkin Vision has received FDA 510(k) clearance for its Eagle device, a Q-switched, 532 nm-wavelength, frequency-doubled Nd:YAG laser developed for selective laser trabeculoplasty (SLT). The Eagle is positioned by Belkin as the first and only contactless laser for glaucoma, offering…

FDA Warms 8 Companies for Unapproved Eye Items
On Monday, the US Food and Drug Administration (FDA) took action by sending warning letters to several companies, including CVS and Walgreens, due to concerns regarding the manufacturing and marketing of unapproved eye products. According to the FDA, these products…

Carl Zeiss Meditec to Acquire D.O.R.C.
Carl Zeiss Meditec AG announced that they have entered into an agreement to acquire 100% of the shares in D.O.R.C. Dutch Ophthalmic Research Center (International) B.V. from Eurazeo SE, based in Paris, France. The deal is worth €985 million (nearly…

RevOpsis Raises $16.5M for Wet AMD Treatment
RevOpsis Therapeutics has announced the successful closure of its first seed funding round, securing a total of $16.5 million. This funding is set to advance the development and commercialization of treatments for chronic multifactorial diseases through the company’s innovative, fully…

LEP Biomedical Gains Funding for HyaGuard Platform
LEP Biomedical announced pre-seed funding of €100,000 ($107,000) over two 6-month periods, which the company says will allow for further preclinical milestones in the development of the HyaGuard platform. The funding was made through Enterprise Ireland. Innovative Design of HyaGuard…

Ocugen Completes Dosing in Key Trial for GA Treatment
Ocugen has officially completed dosing in the second cohort of its phase 1/2 ArMaDa trial of OCU410, a novel gene therapy designed to treat geographic atrophy (GA). The therapy, known as adeno-associated viral vector 5 human RORA (AAV5-hRORA), is being…
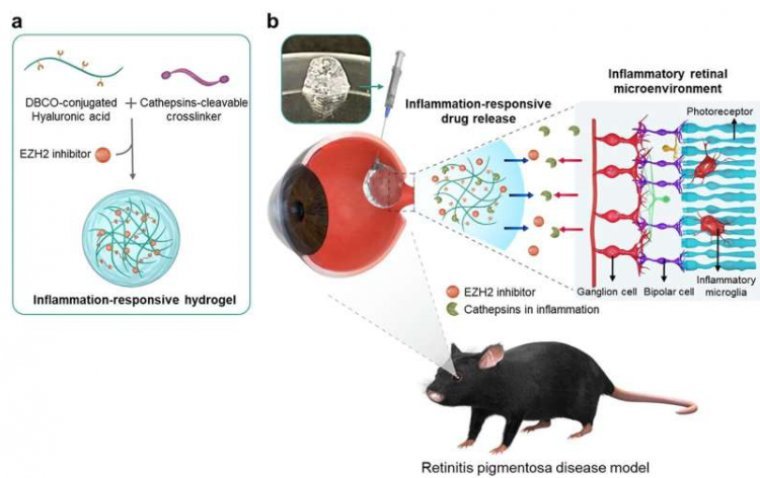
New Treatment Slows Down Progression of AMD, RP
A breakthrough study has unveiled a promising advancement in the treatment of incurable eye diseases such as age-related macular degeneration and retinitis pigmentosa. Researchers have successfully integrated anti-inflammatory drugs into a hydrogel, aiming to combat inflammation in the retina while…

Ocular Therapeutix Reports Promising Axpaxli Results in DR Study
Ocular Therapeutix announced positive topline results from its phase 1 HELIOS study, which evaluated the safety, tolerability, and efficacy of Axpaxli versus a sham control in patients with moderately severe to severe nonproliferative diabetic retinopathy (NPDR) without diabetic macular edema…

Caplin Granted USFDA Approval for Key Ophthalmic Solution
Caplin Steriles Limited (Caplin), a subsidiary of Caplin Point Laboratories Limited, has received final approval from the United States Food and Drug Administration (USFDA) for its Abbreviated New Drug Application (ANDA) for Ketorolac Tromethamine Ophthalmic Solution 0.5%. This product is…





