
Newsletter Subscribe
Enter your email address below and subscribe to our newsletter


Enter your email address below and subscribe to our newsletter
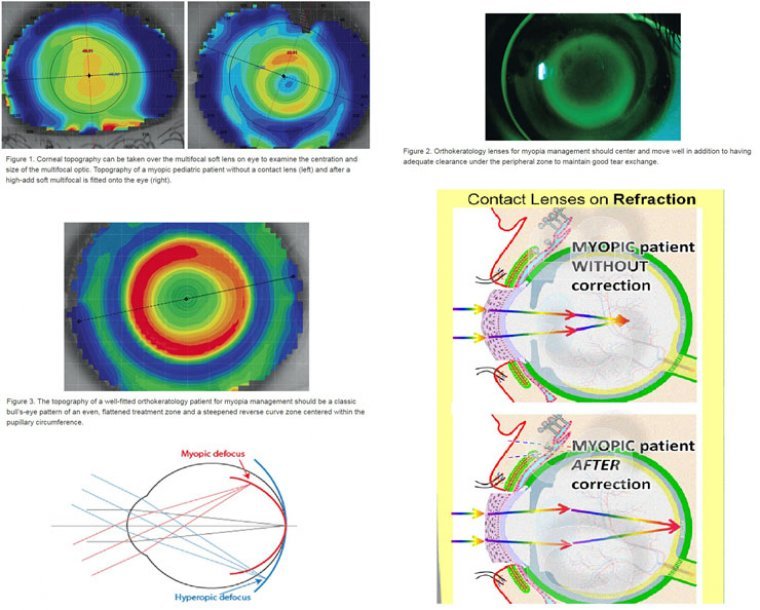
Are you prescribing contact lenses for myopia management? We ask this question at the start of every myopia management lecture we deliver. Over the past two years, we have gone from having audiences with no hands raised to now having…

Nanoscope Therapeutics announced positive topline results from the phase 2b RESTORE trial, evaluating MCO-010, a novel gene therapy aimed at treating severe vision loss caused by advanced retinitis pigmentosa (RP), irrespective of the genetic mutation causing the disease. RESTORE Trial…
Regeneron has received FDA approval for Eylea HD (aflibercept 8 mg) in a significant development anticipated to substantially extend dosing intervals for patients with retinal diseases such as wet age-related macular degeneration (AMD), diabetic macular edema (DME), and diabetic retinopathy (DR).…

Outlook Therapeutics announced that the FDA has issued a complete response letter (CRL) regarding the company’s Biologics License Application (BLA) for ONS-5010 (Lytenava). ONS-5010 is an investigative ophthalmic formulation of bevacizumab aimed at treating wet AMD. While the FDA recognized…
A novel compound, created at the University of Illinois Chicago, holds the potential to provide an alternative to injections for the vast population suffering from an eye condition leading to blindness. This condition, known as wet age-related macular degeneration (AMD),…
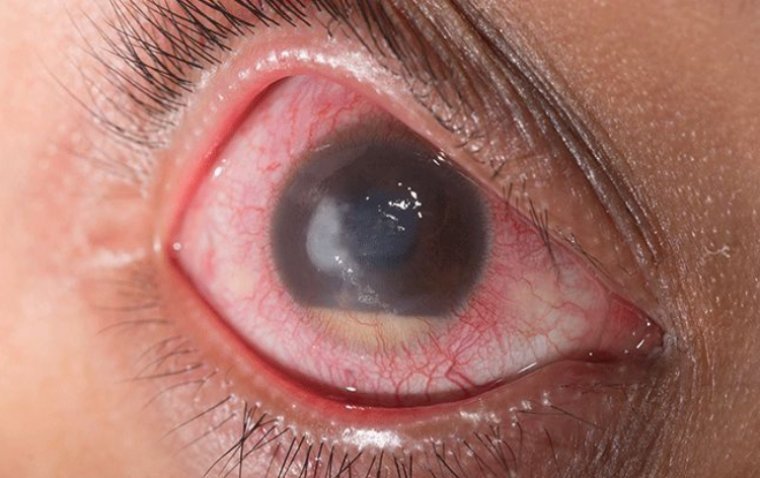
What Is Endophthalmitis? Endophthalmitis is a rare but serious eye condition that can lead to vision loss if not promptly treated. This infection targets the interior structures of the eye, causing inflammation and potential damage. Understanding the symptoms, causes, treatment…

Since February 2023, the FDA and various companies have been actively initiating recalls of eye drops and ointments due to concerns about potential bacterial contamination. The recalls, motivated by the risk of tainted products, have been steadily increasing, underscoring the…

Astellas Pharma has signed a license agreement that grants them the rights to utilize the intravitreal retinotropic R100* vector, developed by 4D Molecular Therapeutics (4DMT). This agreement enables Astellas to target one specific genetic target associated with rare monogenic ophthalmic…
Researchers have successfully developed a groundbreaking synthetic protein to treat macular degeneration and other currently incurable eye diseases leading to blindness in millions of individuals. This pioneering drug, currently undergoing human trials, is the first of its kind. With over…

Pixium Vision SA has announced the reception of a €1.8 million Research Tax Credit from the French government. The funds received will enable the company to extend its cash runway until the end of September 2023, offering greater flexibility to…