
Newsletter Subscribe
Enter your email address below and subscribe to our newsletter


Enter your email address below and subscribe to our newsletter

Skye Bioscience’s phase 2a clinical trial of its SBI-100 ophthalmic emulsion in patients with primary open-angle glaucoma (POAG) or ocular hypertension (OHT) has failed to meet its primary endpoint for lowering intraocular pressure (IOP). Consequently, the company announced its decision…

Melt Pharmaceuticals has completed the dosing of its first patient in its phase 3 program for MELT-300, marking a crucial step in evaluating the safety and efficacy of this pioneering product. MELT-300 is a non-intravenous, non-opioid tablet comprising fixed doses…

Italy’s antitrust regulator has initiated a probe into several pharmaceutical giants, including Novartis, amid suspicions of anti-competitive practices in the sale of a drug targeting eye conditions. The investigation follows concerns regarding potential market restrictions and delayed product launch of…
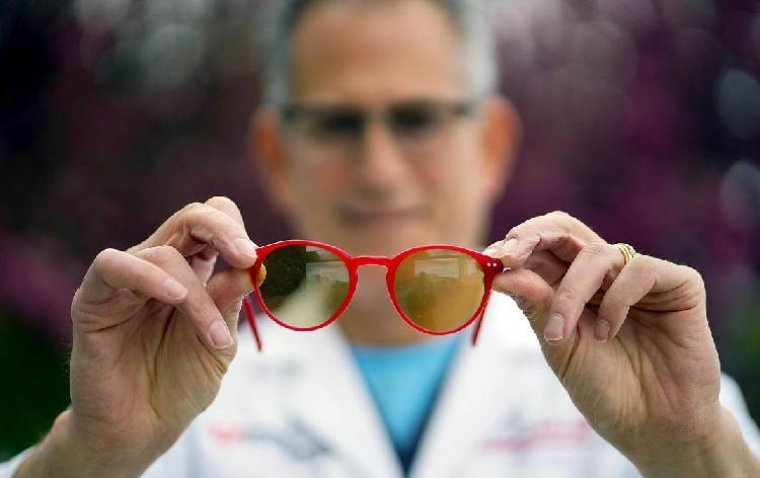
This spring, individuals grappling with migraines find relieve with a novel solution for managing light sensitivity, thanks to new eyeglass lens technology developed by a physician-scientist at the University of Utah John A. Moran Eye Center. The Impact of Migraine…

LambdaVision, a pioneering company dedicated to developing a protein-based artificial retina aimed at restoring sight lost to retinal degenerative diseases, announced the successful first closing of its seed round to advance its artificial retina development. The funding injection will fuel…

Quantel Medical, a division of Lumibird Medical, has unveiled its latest innovation with the launch of the POCKET III handheld pachymeter. This compact device, according to Quantel, sets a new standard as the lightest connected handheld pachymeter on the market,…
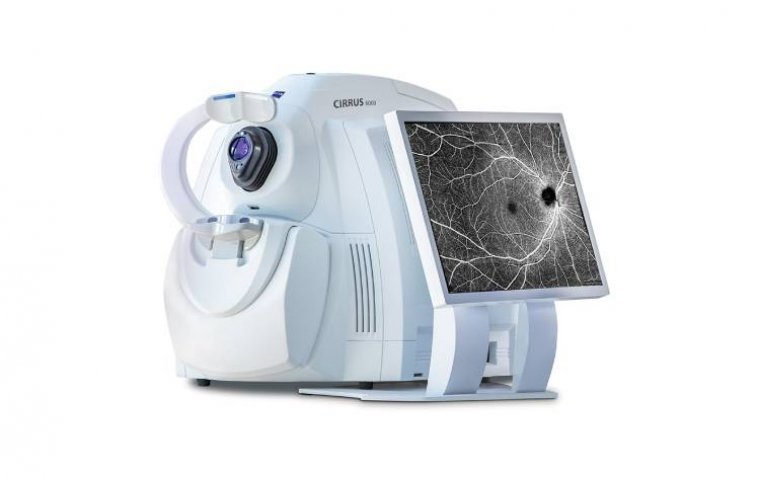
Zeiss Medical Technology has introduced a series of enhancements to the Cirrus 6000, promising a more streamlined and data-centric workflow for ophthalmologists. This update boasts the largest OCT reference database in the US market and fortified cybersecurity features. With recent…

The U.S. Food and Drug Administration (FDA) has qualified the Assessment of IntraOcular Lens Implant Symptoms (AIOLIS), developed by the American Academy of Ophthalmology (AAO), as a Medical Device Development Tool (MDDT). This designation empowers medical device sponsors to employ…

In a significant development for the medical community, Drug Farm announced that the U.S. Food and Drug Administration (FDA) has granted clearance for the Investigational New Drug (IND) application of DF-003. This novel drug, hailed as a first-in-class oral medication,…

Merck & Co, a global pharmaceutical giant, has finalized a monumental deal worth $3 billion to acquire EyeBio, a leading developer of eye-focused drugs. The agreement includes an upfront payment of $1.3 billion in cash, along with an additional $1.7 billion…