Pharma & Drugs
Share your love

Atsena & Nippon Shinyaku Partner on ATSN-101
Atsena Therapeutics, Inc. and Nippon Shinyaku Co., Ltd. have entered an exclusive licensing agreement to advance and commercialize ATSN-101, an investigational gene therapy for Leber congenital amaurosis type 1 (LCA1), in both the United States and Japan. ATSN-101 is designed…
Trial Results for Susvimo Show Sustained Efficacy in DME and DR
Genentech and Roche released compelling 2-year data from the phase 3 Pagoda and Pavilion studies evaluating Susvimo (ranibizumab injection) 100 mg/mL, marking a significant development in the treatment landscape for diabetic macular edema (DME) and diabetic retinopathy (DR). Reintroduction of…

Okyo Pharma’s Phase 2 Trial for Neuropathic Corneal Pain
Okyo Pharma announced its plan to initiate a phase 2 clinical trial for OK-101 in the treatment of neuropathic corneal pain (NCP), scheduled to begin in the third quarter of 2024. This one-year study follows promising results from preclinical animal…

NexThera Submits IND Application for NT-101 for Wet AMD
NexThera has officially submitted a Phase 1/2a Investigational New Drug (IND) application to the U.S. Food and Drug Administration (FDA) for NT-101, an innovative non-invasive eye drop treatment targeting wet age-related macular degeneration (wet AMD). Proprietary Delivery Platform: Advancing Treatment…

ENTOD Pharma Introduces New Eye Drop Preservative System
Mumbai-based ENTOD Pharmaceuticals has unveiled a novel advancement in ophthalmic care with the introduction of EyeBS, a new eye drop preservative system. EyeBS is specifically designed to supplant traditional detergent-based preservatives such as Benzalkonium Chloride (BAK) and Polyquaternium-1, offering a…
Study Highlights Significant Drug Waste from SUCD for Eye Drops
A recent report published online in Ophthalmology reveals alarming levels of drug wastage and associated costs due to self-imposed use cessation dates (SUCD) for multiuse eye drop bottles. John M. Tan and colleagues from the Icahn School of Medicine at…
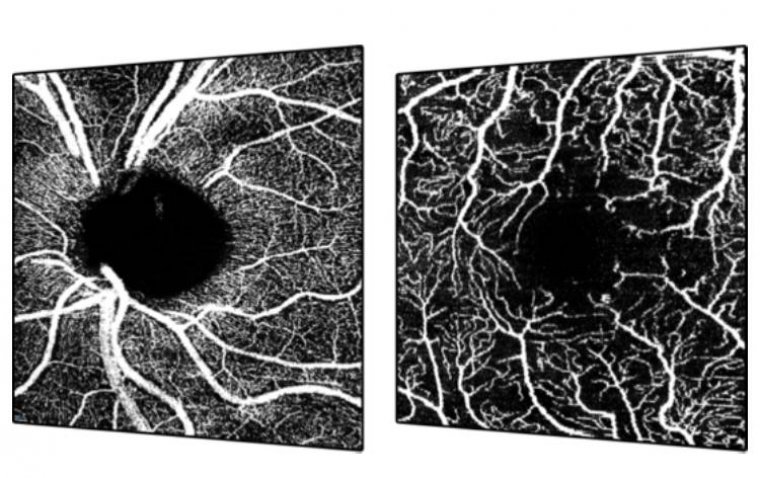
FDA Clears Enhanced SPECTRALIS® OCTA Module by Heidelberg
Heidelberg Engineering announced the FDA clearance of its upgraded SPECTRALIS® OCTA Module featuring SHIFT technology. This advancement promises to significantly reduce acquisition time by 50%, marking a pivotal development in clinical efficiency and diagnostic precision within ophthalmology. Enhancing Clinical Utility…

Sydnexis Appoints Perry J. Sternberg as CEO
Sydnexis, Inc., a pioneering biopharmaceutical company dedicated to addressing pediatric myopia progression, has appointed Perry J. Sternberg as its Chief Executive Officer and as a member of its Board of Directors. Sternberg, a seasoned pharmaceutical and biotech executive with over…

Genentech Reintroduces Susvimo Ocular Implant for Wet AMD
Genentech announced the reintroduction of Susvimo (ranibizumab injection) 100 mg/mL for intravitreal use via ocular implant, aimed at treating wet age-related macular degeneration (AMD). This decision follows the FDA’s approval of a post-approval supplement to the Biologics License Application, which…

Outlook Therapeutics Receives UK Approval of Lytenava for AMD
Outlook Therapeutics announced that the UK Medicines and Healthcare products Regulatory Agency (MHRA) has granted marketing authorization for Lytenava (bevacizumab gamma) for the treatment of wet age-related macular degeneration (AMD) in the UK. Lytenava is the first and only authorized…





