
Newsletter Subscribe
Enter your email address below and subscribe to our newsletter


Enter your email address below and subscribe to our newsletter

4D Molecular Therapeutics (4DMT) announced the FDA clearance of its investigational new drug application (IND) for 4D-175, an R100 vector-based intravitreal genetic medicine intended for treating geographic atrophy (GA) in patients. Phase 1 GAZE Clinical Trial Details The phase 1…
Two recent studies have found that a drug called brolucizumab, which was approved in 2019 and developed by Swiss pharmaceutical company Novartis AG to treat wet age-related macular degeneration (AMD), can cause rare retinal side effects due to its interaction…

DORC International has announced 510(k) market clearance and the launch of the Eva Nexus. According to DORC, the Eva Nexus features multiple innovations to improve performance, efficiency, and stability for retina surgeons, including Smart IOP for anterior and posterior chamber stability; DORC’s trocar cannula system with HI-FLOW infusion…
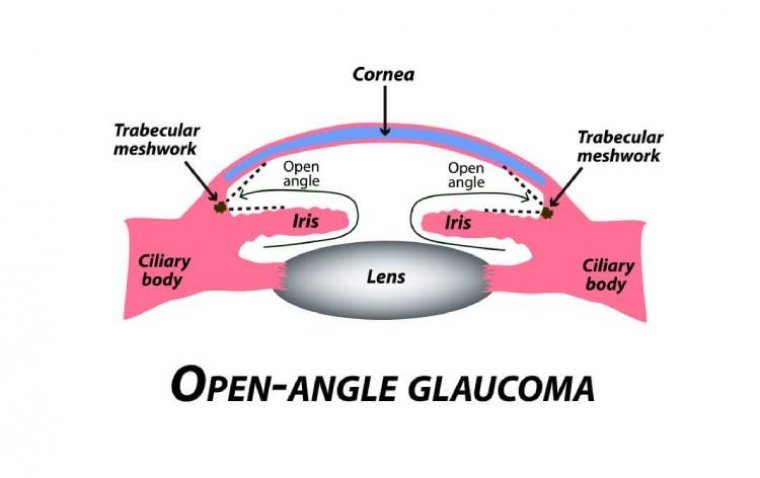
A subtype of glaucomas known as primary open angle glaucoma (POAG) is characterized by an open, seemingly normal anterior chamber angle and elevated intraocular pressure (IOP), with no additional underlying disease. Secondary glaucoma is a condition where the elevated IOP…

Harrow Pharmaceuticals announced its efforts to relaunch Triesence (triamcinolone acetonide injectable suspension) 40 mg/mL in the United States market. The company announced the successful manufacture of the first of three commercial-scale Process Performance Qualification (PPQ) batches required for the relaunch.…

Aurion Biotech has been granted Breakthrough Therapy Designation (BTD) and Regenerative Medicine Advanced Therapy (RMAT) Designation by the FDA for AURN001, Aurion’s innovative allogeneic cell therapy candidate designed to treat corneal edema secondary to corneal endothelial disease. Expediting Development and…

Oculis Holding announced positive topline results from its phase 2b RELIEF trial, shedding light on the efficacy of licaminlimab, an innovative anti-TNFα biologic eye drop, in addressing dry eye disease (DED). Licaminlimab’s established dual mechanism of action, boasting both anti-inflammatory…

In the quest to find effective treatments for eye diseases, the METformin for the MINimization of Geographic Atrophy Progression (METforMIN) trial sought to evaluate the efficacy of oral metformin in slowing the progression of geographic atrophy (GA) and its effects…

Nordic Pharma announced the commercial launch of Lacrifill Canalicular Gel, aimed at addressing dry eye disease in the United States. This novel therapy, Lacrifill, is a cross-linked hyaluronic acid derivative that has received clearance from the FDA. Its primary function…

Johnson & Johnson announced promising topline results from its Phase 3 VIVACITY study evaluating nipocalimab in adults with generalized myasthenia gravis (gMG), alongside findings from the Phase 2 DAHLIAS study in adults with Sjögren’s disease (SjD). Nipocalimab, an investigational treatment,…