
Newsletter Subscribe
Enter your email address below and subscribe to our newsletter


Enter your email address below and subscribe to our newsletter

Glaukos Corporation has confirmed the activation of the CMS Healthcare Common Procedure Coding System’s J-code—J7355—for its iDose TR product. This development marks a significant milestone in enhancing patient access and streamlining coverage and payment processes for the innovative treatment, according…

Formycon AG and its licensing partner Klinge Biopharma GmbH announced the U.S. Food and Drug Administration’s (FDA) approval of FYB203/AHZANTIVE®, a biosimilar to Eylea®. FYB203/AHZANTIVE® has been approved by the FDA for the treatment of patients with Age-Related Neovascular (wet)…
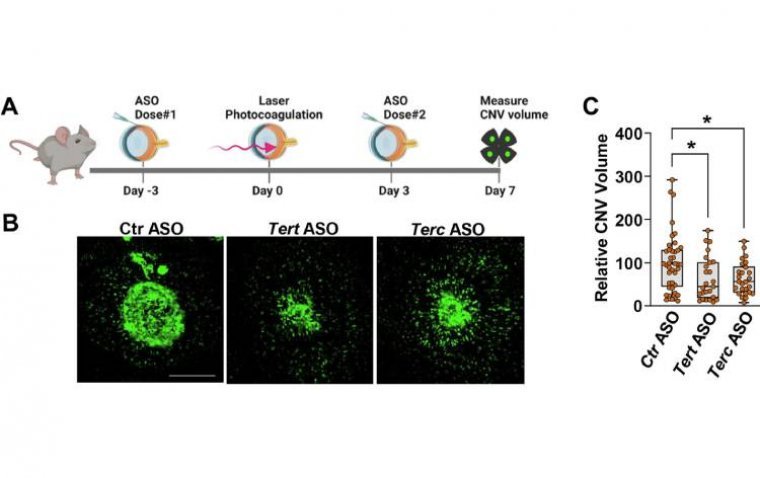
A recent study conducted on mice has provided promising insights into a potential alternative treatment for the “wet” version of age-related macular degeneration (AMD). Researchers identified an enzyme, telomerase, which plays a critical role in the abnormal blood vessel growth…
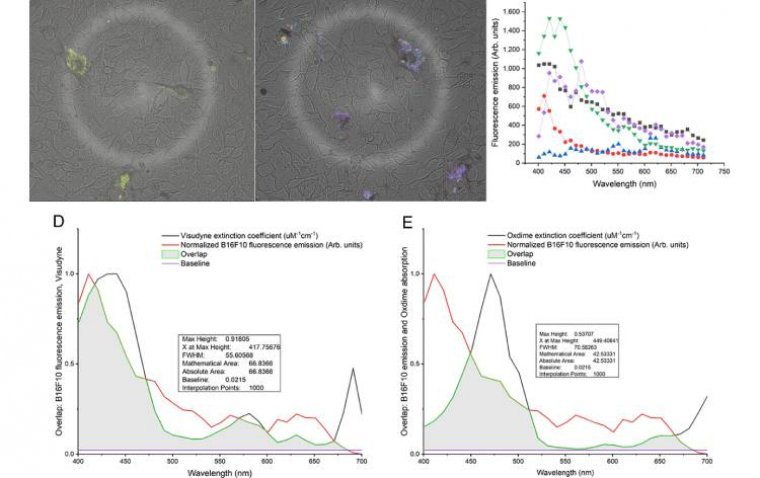
Scientists from the Optics and Photonics Research Center (CePOF) in Brazil, along with collaborators from the University of Toronto and Princess Margaret Cancer Center in Canada, have achieved a significant breakthrough in cancer treatment. Their study, published in Proceedings of…

Alcon has finalized its acquisition of BELKIN Vision, marking a significant expansion of its glaucoma treatment offerings. The acquisition, valued at $81M upfront, with potential additional payments up to $385M contingent on sales milestones, includes BELKIN Vision’s innovative Direct Selective…
Roche announced that the Committee for Medicinal Products for Human Use (CHMP) of the European Medicines Agency (EMA) has issued a positive opinion recommending the extension of marketing authorization for Vabysmo (faricimab). This recommendation includes the treatment of visual impairment…

The Committee for Medicinal Products for Human Use (CHMP) of the European Medicines Agency (EMA) delivered a negative opinion on the marketing authorization application (MAA) for Apellis Pharmaceuticals’ Syfovre (pegcetacoplan), intended for the treatment of geographic atrophy (GA). CHMP’s Assessment…

Thea Pharmaceuticals launched a new website aimed at providing comprehensive resources to ophthalmologists for the effective use of latanoprost ophthalmic solution 0.005%, marketed as Iyuzeh, in patient care. This initiative comes in response to feedback from Eye Care Professionals (ECPs)…

A new artificial intelligence (AI) system developed by researchers from UCL and Moorfields Eye Hospital could potentially revolutionize the recruitment process for clinical trials aimed at treating Geographic Atrophy (GA), an advanced form of dry age-related macular degeneration (AMD) that…

NVISION Eye Centers, a prominent provider of eye care services nationwide, announced a strategic collaboration with Parkhurst NuVision of San Antonio, Texas, through its subsidiary NVision Clinical Research, LLC. This partnership aims to advance clinical trials in ophthalmology drug development,…