
Newsletter Subscribe
Enter your email address below and subscribe to our newsletter


Enter your email address below and subscribe to our newsletter
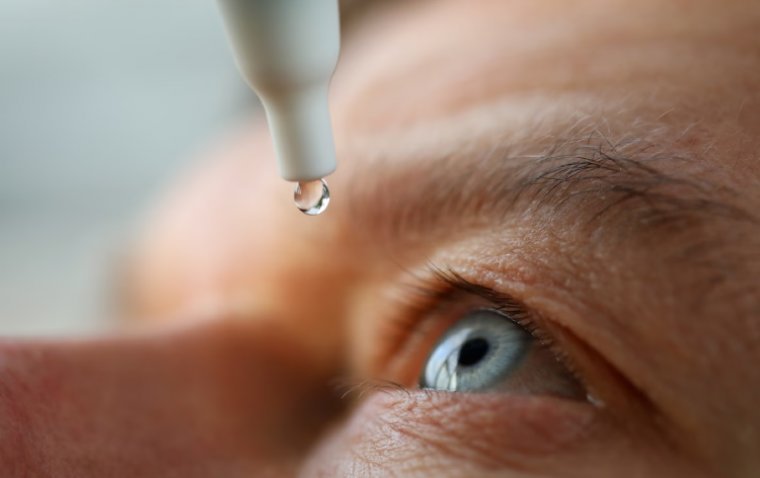
After 30 years of dedicated work, two researchers have achieved a remarkable breakthrough by developing eye drops that can effectively reach the back of the eye. This groundbreaking development paves the way for needle-free treatments of blinding retinal diseases, such…

Eyenovia announced the successful completion of the FDA inspection for its facility in Redwood City, California, marking a significant milestone in the company’s manufacturing capabilities. This achievement signals the green light for the facility to engage in final assembly, packaging,…

Washington University in St. Louis has been granted up to $20 million by the Advanced Research Projects Agency for Health (ARPA-H) to spearhead the development of a novel portable device aimed at revolutionizing the detection of eye diseases. The initiative,…

Ophthalmologists at New York Eye and Ear Infirmary of Mount Sinai (NYEE) have pioneered a novel protocol aimed at swiftly diagnosing eye stroke and expediting care to prevent irreversible vision loss. Published in Ophthalmology, their study outlines the use of…
Google DeepMind’s gemini AI versus ChatGPT: a comparative analysis in ophthalmology | Eye Nature Source: Author: | Date: 2024-02-14 08:00:00 Source: Author: | Date: 2024-02-14 08:00:00

A recent study published online in JAMA Ophthalmology sheds light on the significant impact of home environment, particularly lighting, on the activity levels of older adults with visual impairment. Conducted by Seema Banerjee, Ph.D., and colleagues from Johns Hopkins University,…

In 2023, the ophthalmic industry witnessed a transformative landscape shaped by strategic company acquisitions, setting the stage for significant advancements and collaborations. The top 10 acquisitions of the year highlighted a dynamic shift in the competitive dynamics of the sector.…

Meridian Medical, headquartered in Switzerland, announced the launch of its line of ophthalmic lasers in the United States, following a series of FDA approvals. The FDA approval, received on January 25, includes the MR Q family of lasers, which offer…

Nicox SA announced an agreement with Kowa, granting the latter exclusive rights in Japan for the development and commercialization of NCX 470, a promising nitric oxide (NO)-donating bimatoprost eye drop designed for reducing intraocular pressure (IOP) in individuals with glaucoma…

Okyo Pharma’s first investigational new drug (IND) application for OK-101 has been cleared by the U.S. Food and Drug Administration (FDA), marking a pivotal moment in the development of potential therapies for neuropathic corneal pain. Initially proposed as an open-label…