
Newsletter Subscribe
Enter your email address below and subscribe to our newsletter


Enter your email address below and subscribe to our newsletter

AEYE Health has received FDA clearance for its fully autonomous AI, making it the first to diagnose referable diabetic retinopathy using a handheld camera. The technology, known as AEYE Diagnostic Screening (AEYE-DS), was previously approved for use with a tabletop…

Inflammasome Therapeutics has officially dosed the first patient in a pioneering clinical trial aimed at treating geographic atrophy (GA) resulting from age-related macular degeneration (AMD). This trial, a first-in-class study for a sustained release implant, is sponsored by the University…
OpenAI’s Sora in ophthalmology: revolutionary generative AI in eye health Nature Source: Author: | Date: 2024-04-30 07:00:00 Source: Author: | Date: 2024-04-30 07:00:00

TED is an autoimmune disease in which the eye muscles and fatty tissue behind the eye become inflamed. This inflammation can push the eyes forward (“staring” or “bulging”) or cause the eyes and eyelids to become red and swollen. In…
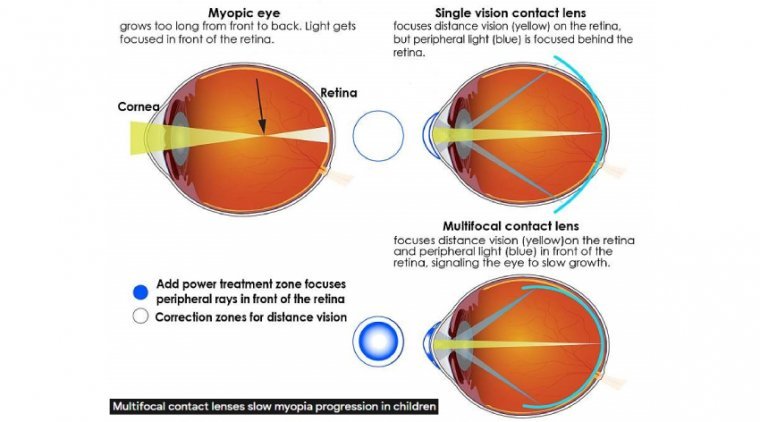
Myopia, or short-sightedness, is one of the most common eye health issues in children and means that they can’t see objects that are far away, causing their vision to blur. Symptoms usually appear between the ages of 8 and 12…
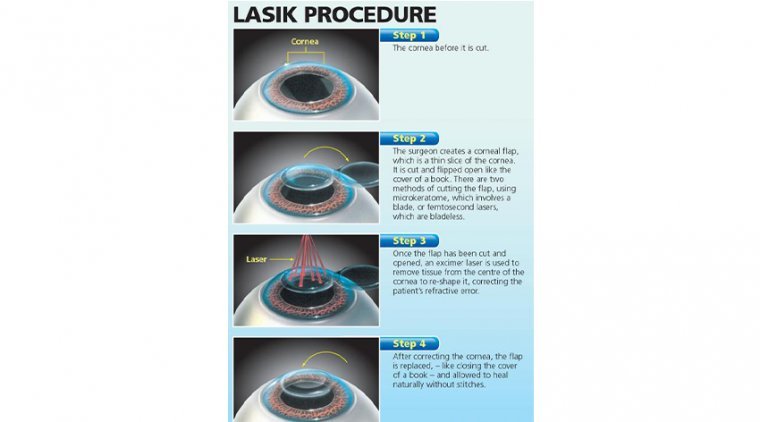
In the field of ophthalmology, laser technology is used in many basic and clinical disciplines and specialities. It has played an important role in promoting the development of ophthalmology. Advancements in technology have allowed improvements in the surgical safety, efficacy, speed…
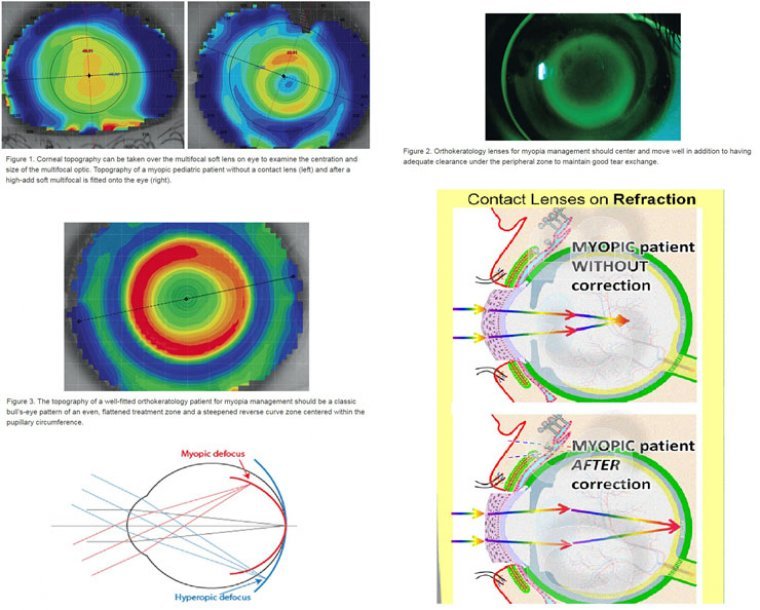
Are you prescribing contact lenses for myopia management? We ask this question at the start of every myopia management lecture we deliver. Over the past two years, we have gone from having audiences with no hands raised to now having…

Nanoscope Therapeutics announced positive topline results from the phase 2b RESTORE trial, evaluating MCO-010, a novel gene therapy aimed at treating severe vision loss caused by advanced retinitis pigmentosa (RP), irrespective of the genetic mutation causing the disease. RESTORE Trial…
Regeneron has received FDA approval for Eylea HD (aflibercept 8 mg) in a significant development anticipated to substantially extend dosing intervals for patients with retinal diseases such as wet age-related macular degeneration (AMD), diabetic macular edema (DME), and diabetic retinopathy (DR).…

Outlook Therapeutics announced that the FDA has issued a complete response letter (CRL) regarding the company’s Biologics License Application (BLA) for ONS-5010 (Lytenava). ONS-5010 is an investigative ophthalmic formulation of bevacizumab aimed at treating wet AMD. While the FDA recognized…