
Newsletter Subscribe
Enter your email address below and subscribe to our newsletter


Enter your email address below and subscribe to our newsletter

Innovent Biologics announced that its second Phase 2 clinical trial of efdamrofusp alfa (R&D code: IBI302), a high-dose recombinant human VEGFR-Fc-Human CR1 fusion protein injection, has achieved its primary endpoint in treating Chinese subjects with neovascular age-related macular degeneration (nAMD).…
In a groundbreaking era of medical research, scientists and clinicians are spearheading novel gene therapies that hold promise for transforming the landscape of eye disease treatment. These cutting-edge interventions address a spectrum of conditions ranging from Usher Syndrome and retinoschisis…

OcuTerra Therapeutics announced the topline results from its phase 2 DR:EAM (Diabetic Retinopathy: Early Active Management) clinical trial, evaluating the efficacy of nesvategrast (OTT166). Nesvategrast, an innovative RGD integrin inhibitor formulated as an eye drop, was under investigation for its…

MediPrint Ophthalmics has successfully completed its SIGHT-2 phase 2b study, showcasing the efficacy of its innovative product, LL-BMT1, against a control group using bimatoprost 0.01% ophthalmic solution. LL-BMT1, a preservative-free, weekly drug-eluting contact lens, utilizes the company’s 3D printing technology,…

Innovent Biologics announced the successful achievement of the primary endpoint in the phase 3 registrational study, RESTORE-1, of IBI311 in China. This recombinant anti-insulin-like growth factor 1 receptor (IGF-1R) antibody marks a pivotal advancement in the treatment of thyroid eye…

Eversight, a leading non-profit dedicated to the restoration of sight through the recovery, processing, and distribution of corneal tissue, announced a strategic partnership with Emmecell Biotech, a biotechnology company focused on developing novel cell therapies for ocular diseases. This collaboration…

Opthea Limited announced Dr. Arshad M. Khanani, MD, MA, FASRS, as its new Chief Medical Advisor. Dr. Khanani, renowned in retina specialization and clinical research, brings a wealth of expertise from his tenure at Sierra Eye Associates and his academic…

Opthea has reached a significant milestone in its quest to combat wet age-related macular degeneration (AMD) with the completion of patient enrollment in the COAST phase 3 pivotal clinical trial. The trial investigates sozinibercept (OPT-302), a promising vascular endothelial growth…
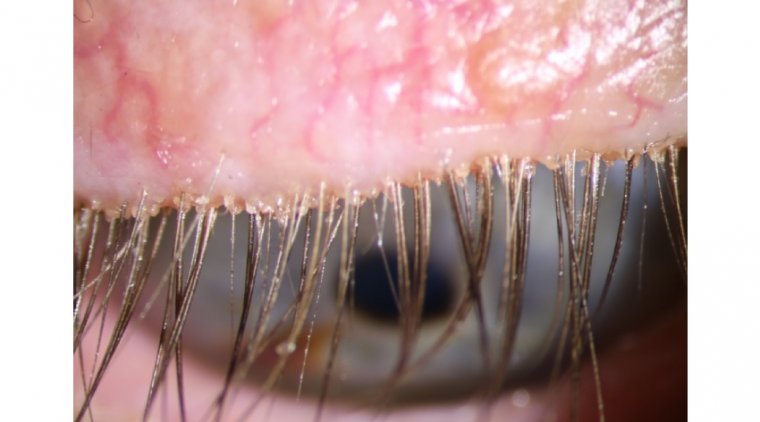
A team of investigators, led by Elizabeth Yeu, MD, from Virginia Eye Consultants in Norfolk, Virginia, reported the safety and efficacy of lotilaner ophthalmic solution, 0.25% (TP-03, Tarsus Pharmaceuticals Inc.), in treating Demodex blepharitis. Demodex mites are the most common…

Swiss-based pharmaceutical company Roche has announced the discontinuation of its oral diabetic retinopathy treatment, vicasinabin, after successfully completing Phase II clinical trials. In a recent presentation to investors, Roche confirmed its decision to halt further investment in vicasinabin, a promising…