
Newsletter Subscribe
Enter your email address below and subscribe to our newsletter


Enter your email address below and subscribe to our newsletter
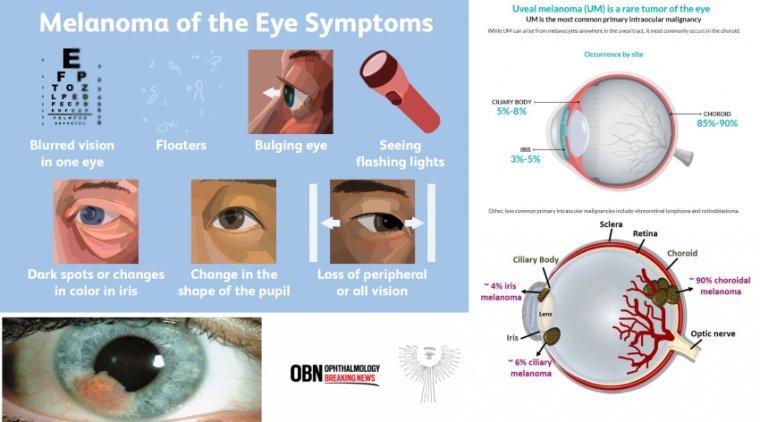
Melanoma of the uveal tract (iris, ciliary body, and choroid), though rare, is the most common primary intraocular malignancy in adults. The mean age-adjusted incidence of uveal melanoma in the United States is approximately 4.3 new cases per million people,…
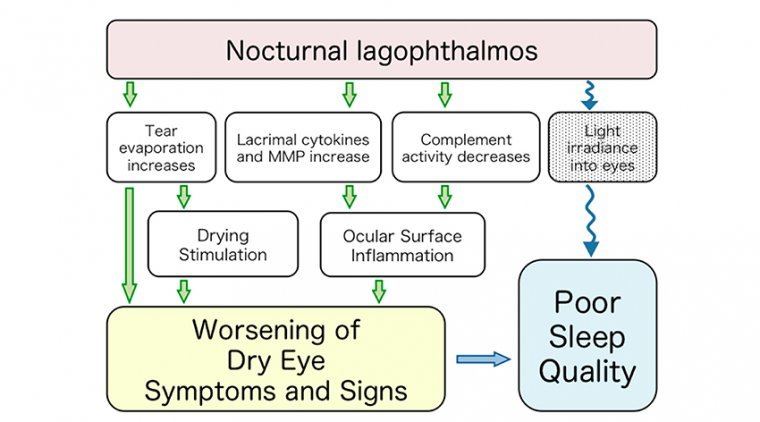
Nocturnal lagophthalmos is the inability to close the eyelids while sleeping. This can result in a range of conditions from mild epithelial changes to severe exposure ulcers (Katz and Kaufman, 1977). Lagophthalmos is associated with exposure keratopathy, poor sleep, and…

4D Molecular Therapeutics (4DMT) announced positive interim data from the Phase 2 PRISM clinical trial, marking a significant stride in the treatment of wet age-related macular degeneration (wet AMD). Arshad M. Khanani, MD, MA, FASRS, presented the data at the…

Kiora Pharmaceuticals has joined forces with Théa Open Innovation (TOI), a sister company of Laboratoires Théa, to develop and commercialize KIO-301 for the treatment of degenerative retinal diseases. The strategic agreement grants Théa exclusive worldwide development and commercialization rights, excluding…

UMass Chan Medical School has awarded exclusive global rights for the development and commercialization of innovative adeno-associated virus (AAV) gene therapy products targeting Stargardt disease and Leber congenital amaurosis type 10 (LCA10) to Iveric Bio, an Astellas Company. While gene…

A cautionary note has been sounded by a researcher from the University of Houston’s College of Optometry regarding the use of low-level red light (LLRL) therapy for controlling myopia, particularly in children. Despite recent studies touting its effectiveness in reducing…

Dr. Reginald J. Sanders, a distinguished physician affiliated with the Retina Group of Washington (RGW) and PRISM Vision Group, has been named as the new president of the American Society of Retina Specialists (ASRS). Dr. Sanders holds the distinction of…

Mireca Medicines has been awarded a $989,000 ‘Translational Research Acceleration Program Award’ from the Foundation Fighting Blindness, enabling the company to advance preclinical development of its lead product, MM238. Recognizing Mireca’s pivotal role in targeting the cGMP pathway for treating…

Formosa Pharmaceuticals announced a strategic licensing agreement with Cristália Produtos Químicos Farmacêuticos to secure exclusive commercialization rights for APP13007 (clobetasol propionate ophthalmic nanosuspension, 0.05%) in Brazil. APP13007 is designed for the treatment of inflammation and pain following ocular surgery and…

Ocular Therapeutix announced that the FDA has granted approval for a modification to the special protocol assessment (SPA) agreement for the pivotal phase 3 SOL clinical trial of Axpaxli (axitinib intravitreal implant, also known as OTX-TKI). This modification marks a…