
Newsletter Subscribe
Enter your email address below and subscribe to our newsletter


Enter your email address below and subscribe to our newsletter

Harrow, a leading pharmaceutical company, has revealed the encouraging results from its ESSENCE‑2 open-label extension (OLE) clinical study for Vevye (cyclosporine ophthalmic solution) 0.1%, a novel treatment for dry eye disease (DED). Study Design and Outcomes: ESSENCE-2 OLE Analysis The…

Italy’s antitrust regulator has initiated a probe into several pharmaceutical giants, including Novartis, amid suspicions of anti-competitive practices in the sale of a drug targeting eye conditions. The investigation follows concerns regarding potential market restrictions and delayed product launch of…

Eversight has partnered up with LighTopTech to elevate the standards of imaging modalities for eye tissues. This ambitious partnership, spanning over four years, sets out to redefine the landscape of ocular imaging in both clinical and research domains. LighTopTech’s OCX…

CorneaGen, a leading provider of ocular tissue, announced the commercial launch of Corneal Tissue Addition for Keratoplasty (CTAK), a novel solution for corneal contouring in keratoconic eyes. Developed in collaboration with CTAK LLC and Ziemer, CTAK offers patient-ready, gamma-irradiated sterile…

BIOPHTA, a preclinical biotech company, has successfully closed a Seed round, raising €6.5 million to advance its pioneering technology into clinical development for the treatment of glaucoma and macular edema. This significant funding injection, led by prominent European venture capital…

Azura Ophthalmics has reached a significant milestone with the enrollment of the first patient in the ASTRO study—a phase 3 clinical trial evaluating the efficacy and safety of AZR-MD-001 in patients grappling with clinical signs and symptoms of meibomian gland…
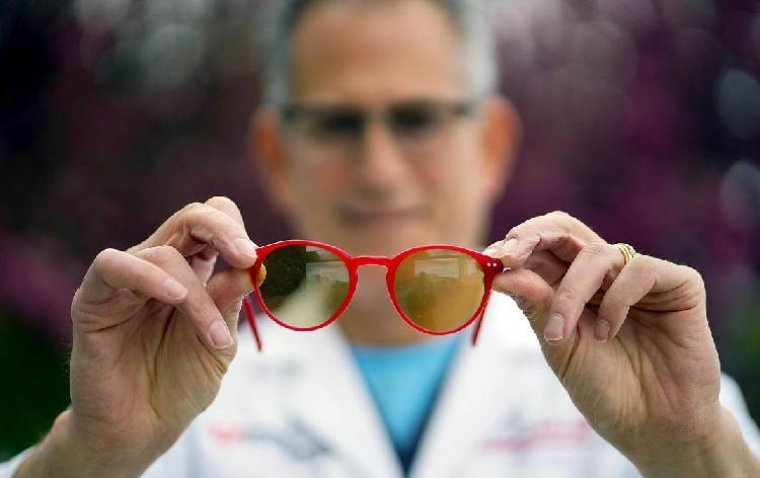
This spring, individuals grappling with migraines find relieve with a novel solution for managing light sensitivity, thanks to new eyeglass lens technology developed by a physician-scientist at the University of Utah John A. Moran Eye Center. The Impact of Migraine…
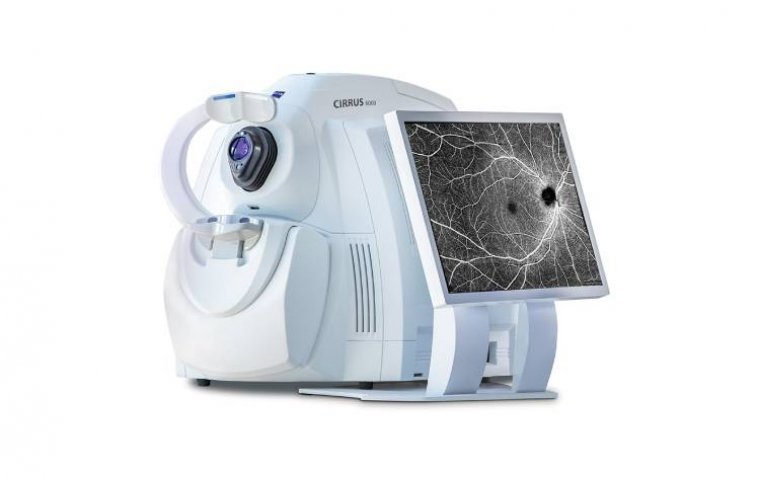
Zeiss Medical Technology has introduced a series of enhancements to the Cirrus 6000, promising a more streamlined and data-centric workflow for ophthalmologists. This update boasts the largest OCT reference database in the US market and fortified cybersecurity features. With recent…

In a significant development for the medical community, Drug Farm announced that the U.S. Food and Drug Administration (FDA) has granted clearance for the Investigational New Drug (IND) application of DF-003. This novel drug, hailed as a first-in-class oral medication,…
The USC Roski Eye Institute is making significant strides in the realm of Thyroid Eye Disease (TED) treatment through pioneering clinical trials exploring new medications. TED, a condition characterized by bulging eyes and potential vision impairment, has traditionally necessitated multiple…