
Newsletter Subscribe
Enter your email address below and subscribe to our newsletter


Enter your email address below and subscribe to our newsletter

Biocon Biologics today announced that the US FDA has approved the Company’s first-to-file application for Yesafili™ (aflibercept-jbvf), an interchangeable biosimilar aflibercept. Indications and Uses of YESAFILI YESAFILI, a vascular endothelial growth factor (VEGF) inhibitor used to treat several ophthalmology conditions,…

Researchers at the University of East Anglia (UEA) have developed a novel resin suitable for 3D printing intraocular devices. This pioneering innovation holds the potential to revolutionize the production of eye implants, which are crucial for cataract and refractive surgeries,…
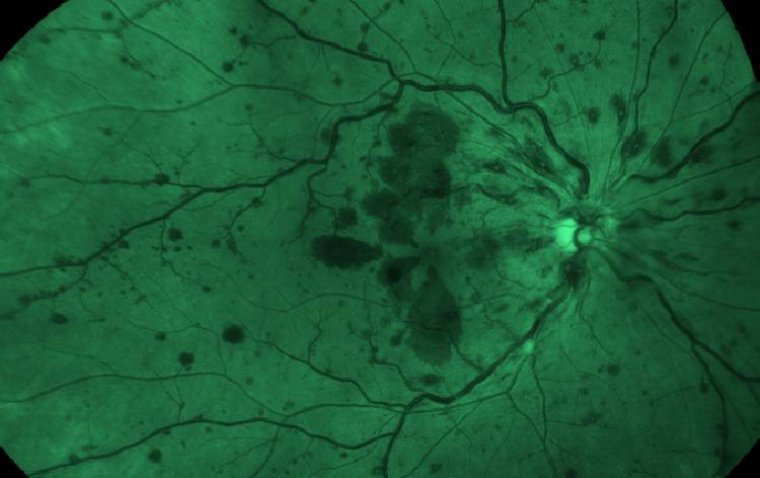
Dr. Fu-Shin Yu, a professor of Ophthalmology, Visual and Anatomical Sciences at Wayne State University School of Medicine, has been awarded a significant grant from the National Eye Institute of the National Institutes of Health. The five-year grant, totaling $2,167,882,…

Glenmark Pharmaceuticals has received final approval by the FDA for Brimonidine Tartrate and Timolol Maleate Ophthalmic Solution, 0.2%|0.5% for glaucoma treatment. This solution has been recognized by the FDA as bioequivalent and therapeutically equivalent to Combigan® Ophthalmic Solution, 0.2%|0.5% by…

A study led by a University at Buffalo researcher has the potential to revolutionize the treatment of retinopathy of prematurity (ROP), a condition that can lead to blindness in low birthweight premature infants if left untreated. Published in eClinicalMedicine, the…
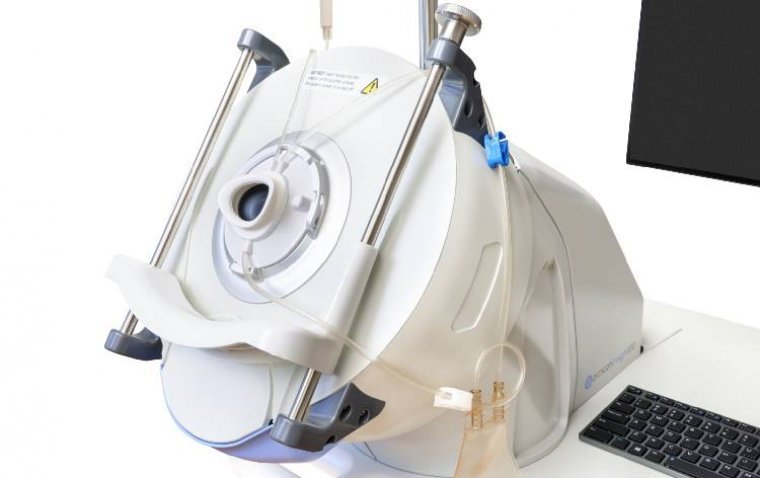
ArcScan, a global medical technology company dedicated to revolutionizing ophthalmic care, marked a significant milestone with the announcement of the National Medical Products Administration (NMPA) approval of its flagship product, the ArcScan Insight® 100, for use in China. Addressing the…

EyeCRO recently announced the results from the inaugural clinical application of its MiDROPS (Microemulsion Drug Ocular Penetration System) technology, presented by Telios Pharma at the Association for Research in Vision and Ophthalmology (ARVO) meeting held in Seattle, WA. The presentation,…

Researchers have developed a prototype “smart” contact lens that accurately measures eye pressure regardless of temperature, according to a study in ACS Applied Materials & Interfaces. Around 3 million people in the U.S. suffer from glaucoma, according to the Centers…

Retinopathy of Prematurity is one of the critical challenges in neonatal care, especially for premature babies born before 31 weeks of gestation and known as grams weighing less than 1,500. This disease, expressing itself as impaired or even complete abnormalities in…

Oculis has completed enrollment in its multicenter, randomized, double-blind, placebo-controlled phase 2 ACUITY trial evaluating the safety and tolerability of OCS-05 in patients with Acute Optic Neuritis (AON). Topline results are expected in Q4 2024. The phase 2 ACUITY study…