
Newsletter Subscribe
Enter your email address below and subscribe to our newsletter


Enter your email address below and subscribe to our newsletter
A novel multi-ingredient supplement significantly improves ocular symptom severity and tear production in patients with dry eye disease: results from a randomized, placebo-controlled clinical trial Frontiers Source: Author: | Date: 2024-04-23 07:00:00 Source: Author: | Date: 2024-04-23 07:00:00

Aldeyra Therapeutics announced its clinical development strategy aimed at the resubmission of a New Drug Application (NDA) for its topical ocular 0.25% reproxalap. This investigational RASP modulator is poised for the treatment of dry eye disease, pending approval from the…

Aurion Biotech has dosed the first Canadian participant in its phase 1/2 clinical trial, known as ABA-1, CLARA. The trial investigates the efficacy and safety of AURN001, a novel cell therapy aimed at treating corneal edema resulting from corneal endothelial…

Opus Genetics recently completed the first stage of its innovative clinical trial, focusing on a new treatment for patients suffering from Leber congenital amaurosis (LCA) due to biallelic mutations in the LCA5 gene. The trial, which is in the phase…

BlueRock Therapeutics LP, a clinical-stage cell therapy company and a subsidiary of Bayer, in partnership with Foundation Fighting Blindness, has announced their collaboration to introduce a new cohort to the Foundation’s ‘Uni-Rare natural history study’. This initiative focuses on individuals…
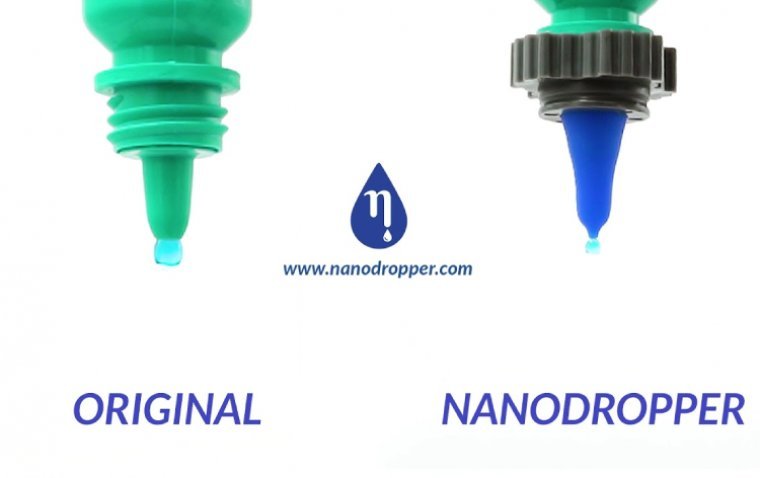
In a pivotal development in the field of ophthalmology, Nanodropper Inc announced that the results from a clinical trial evaluating the Nanodropper® Adaptor have been published in the esteemed journal, Ophthalmology. The study’s findings indicate that there was no clinically…

In a recent phase II clinical trial conducted by the National Eye Institute (NEI), part of the National Institutes of Health, the antibiotic minocycline, known for its anti-inflammatory properties, was tested for its efficacy in slowing down the progression of…

Innovent Biologics announced that its second Phase 2 clinical trial of efdamrofusp alfa (R&D code: IBI302), a high-dose recombinant human VEGFR-Fc-Human CR1 fusion protein injection, has achieved its primary endpoint in treating Chinese subjects with neovascular age-related macular degeneration (nAMD).…
In a groundbreaking era of medical research, scientists and clinicians are spearheading novel gene therapies that hold promise for transforming the landscape of eye disease treatment. These cutting-edge interventions address a spectrum of conditions ranging from Usher Syndrome and retinoschisis…

OcuTerra Therapeutics announced the topline results from its phase 2 DR:EAM (Diabetic Retinopathy: Early Active Management) clinical trial, evaluating the efficacy of nesvategrast (OTT166). Nesvategrast, an innovative RGD integrin inhibitor formulated as an eye drop, was under investigation for its…