
Newsletter Subscribe
Enter your email address below and subscribe to our newsletter


Enter your email address below and subscribe to our newsletter

Aldeyra Therapeutics announced the receipt of a Complete Response Letter from the U.S. Food and Drug Administration (FDA) in response to the New Drug Application (NDA) for reproxalap, an investigational drug candidate designed for the treatment of dry eye disease.…
In children with amblyopia, often referred to as “lazy eye,” one eye exhibits weaker vision than the other due to various factors. This weaker eye may struggle with focusing, misalignment due to strabismus, or vision obstruction from conditions like cataracts…

The Sir Jules Thorn Charitable Trust has allocated £5 million towards the establishment of a research center within Oriel, the collaborative initiative between UCL and Moorfields Eye Hospital aimed at advancing eye health. Partnering with Moorfields Eye Charity and Moorfields…
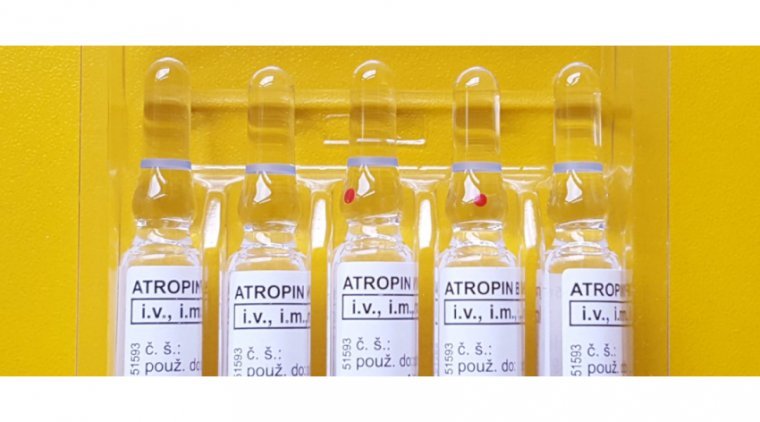
According to a report by an international team of researchers, atropine instillation after trabeculectomy should be discouraged since it causes greater and longer lasting reduction in visual quality.1 The study’s lead author is Panagiotis Laspas, MD, of the Department of…
According to recent research, perfluorohexyloctane eye drops (SHR8058) demonstrated significant improvement in both the signs and symptoms of dry eye disease (DED) linked to meibomian gland dysfunction (MGD) with rapid effectiveness in Chinese patients. The findings showed that the perfluorohexyloctane…

4D Molecular Therapeutics (4DMT) announced positive interim data from the Phase 2 PRISM clinical trial, marking a significant stride in the treatment of wet age-related macular degeneration (wet AMD). Arshad M. Khanani, MD, MA, FASRS, presented the data at the…

Kiora Pharmaceuticals has joined forces with Théa Open Innovation (TOI), a sister company of Laboratoires Théa, to develop and commercialize KIO-301 for the treatment of degenerative retinal diseases. The strategic agreement grants Théa exclusive worldwide development and commercialization rights, excluding…

Ocular Therapeutix announced that the FDA has granted approval for a modification to the special protocol assessment (SPA) agreement for the pivotal phase 3 SOL clinical trial of Axpaxli (axitinib intravitreal implant, also known as OTX-TKI). This modification marks a…

In a significant stride towards advancing its clinical development program for ONS-5010, Outlook Therapeutics has received written agreement from the FDA under a special protocol assessment (SPA) for the NORSE EIGHT clinical trial protocol. This trial aims to evaluate the…

Aviceda Therapeutics announced a significant milestone in the development of its lead ophthalmic candidate, AVD-104, with the enrollment of the first patient in the GLYCO phase 2 US clinical trial. The trial aims to evaluate the efficacy and safety of…