
Newsletter Subscribe
Enter your email address below and subscribe to our newsletter


Enter your email address below and subscribe to our newsletter

Ocular Therapeutix has obtained written FDA approval for the comprehensive design of its pivotal phase 3 clinical trial, known as the SOL trial, which evaluates Axpaxli (axitinib intravitreal implant) for treating wet age-related macular degeneration (AMD) under a Special Protocol…

OnPoint Vision has officially received FDA approval for their investigational device exemption (IDE) application, initiating the phase 1 in the pivotal clinical trial for the AccuraSee Intraocular Pseudophakic Capsular Lens (IOPCL) Magnifier (MAG). This trial focuses on the secondary implantation…

At the J.P. Morgan Healthcare Conference, Novartis CEO Vas Narasimhan denied rumors that the pharmaceutical giant is planning to sell its respiratory and ophthalmology units altogether. The rumors, which have been circulating for several weeks, suggested that Novartis was looking…

According to sources familiar with the matter, Novartis AG is kicking off the process of selling some of its ophthalmology assets in order to prune its portfolio and concentrate on other therapeutic areas, including hematology, immunology, solid tumors, neurology, and…

Bausch + Lomb has announced that it will acquire the dry eye drug Xiidra (lifitegrast ophthalmic solution) 5% from Novartis. Xiidra is an eye drop without steroids that has received specific approval for treating the signs and symptoms of dry…
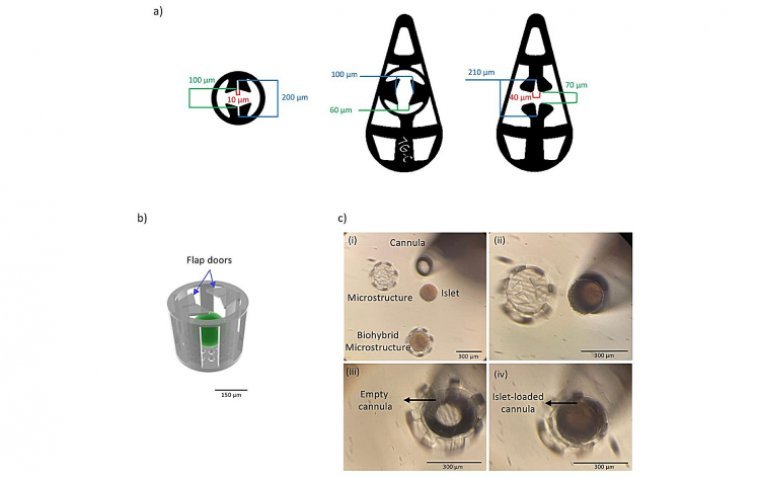
In Sweden, researchers have created a microscale device designed for implantation in the eye, opening up new paths for cell-based treatments for conditions like diabetes. Developed by a team from KTH Royal Institute of Technology and Karolinska Institutet, this 3D-printed…

Ocuphire Pharma and Viatris announced that the FDA has granted approval for Ryzumvi (phentolamine ophthalmic solution) 0.75%. This approval specifically authorizes its use in the treatment of pharmacologically-induced mydriasis, produced by adrenergic agonists (such as phenylephrine) or parasympatholytic agents (like…

Thea Pharma announced the launch and availability of Iyuzeh (latanoprost ophthalmic solution) 0.005% in the United States market. Iyuzeh, having obtained FDA approval in December 2022, stands as the first preservative-free latanoprost product for individuals in the US diagnosed with…
Bimatoprost is a medicine that helps with high pressure in the eye (OCT) and open-angle glaucoma. When too much fluid builds up in your eyes the pressure in the eye increases. Bimatoprost drops help to drain the excess fluid, and…
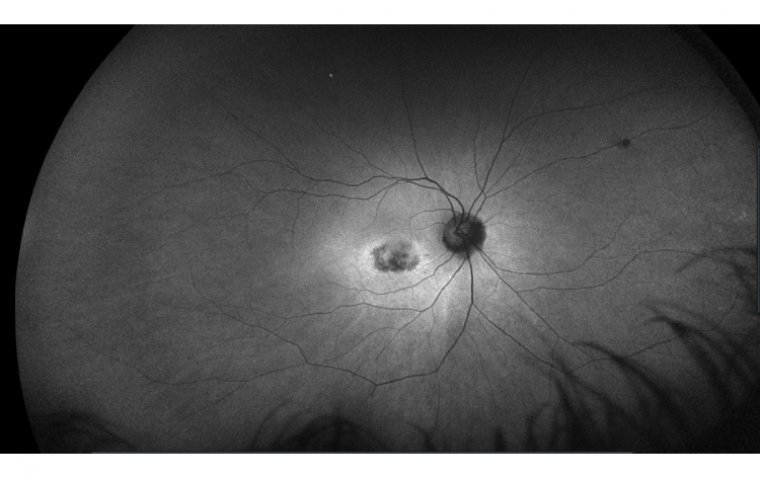
What Is Plaquenil Retinal Toxicity? Plaquenil (hydroxychloroquine) is a medication used to treat autoimmune diseases such as lupus and rheumatoid arthritis. However, in rare cases, it can cause damage to the retina, a layer of tissue at the back of…